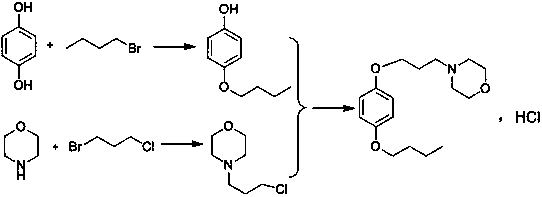
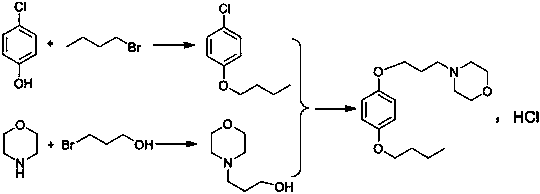
A kind of preparation method of pramoxine hydrochloride
A technology of pramoxine hydrochloride and potassium carbonate, which is applied in the field of pharmaceutical chemical synthesis, can solve problems such as dark color, achieve high reaction yield, less impurities, and avoid dyeing
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] (1) Preparation of 4-n-butoxychlorobenzene
[0029] Add 771g of water, 84g of sodium hydroxide and 328.8g of n-bromobutane to the reaction flask, pass nitrogen protection, then add 257g of p-chlorophenol; the reaction temperature is slowly raised at 70-75°C for 15 hours. After the reaction was completed, the solution was cooled to 5-10°C and stirred for 1 hour, filtered, and the obtained off-white solid was beaten and washed with 650 g of water for half an hour, filtered again, and dried to obtain 317.6 g of white 4-n-butoxychlorobenzene; the molar yield was 86 %, the liquid phase purity is 99.52%.
[0030] (2) Preparation of N-(3-hydroxypropyl)morpholine
[0031] Add 609g of toluene, 174g of morpholine and 333.6g of 3-bromopropanol into the reaction flask, add 276g of potassium carbonate while stirring, and react at 55-60°C for 15 hours. After the reaction was completed, press filtration, and the filtrate was concentrated to remove toluene and unreacted raw materials...
Embodiment 2
[0035] (1) Preparation of 4-n-butoxychlorobenzene
[0036] Add 771g of water, 88g of sodium hydroxide and 356.2g of n-bromobutane to the reaction bottle, pass nitrogen protection, then add 257g of p-chlorophenol; the reaction temperature is slowly raised at 90-95°C for 10 hours. After the reaction, the solution was cooled to 5-10°C and stirred for 1 hour, filtered, and the obtained off-white solid was beaten and washed with 650 g of water for half an hour, filtered again, and dried to obtain 319.5 g of white 4-n-butoxychlorobenzene; the molar yield was 86.5 %, the liquid phase purity is 99.51%.
[0037] (2) Preparation of N-(3-hydroxypropyl)morpholine
[0038] Add 913.5 g of toluene, 174 g of morpholine and 556 g of 3-bromopropanol into the reaction flask, add 331.2 g of potassium carbonate while stirring, and react at 75-80° C. for 8 hours. After the reaction was completed, press-filtered, and the filtrate was concentrated to remove toluene and unreacted raw materials to ob...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More