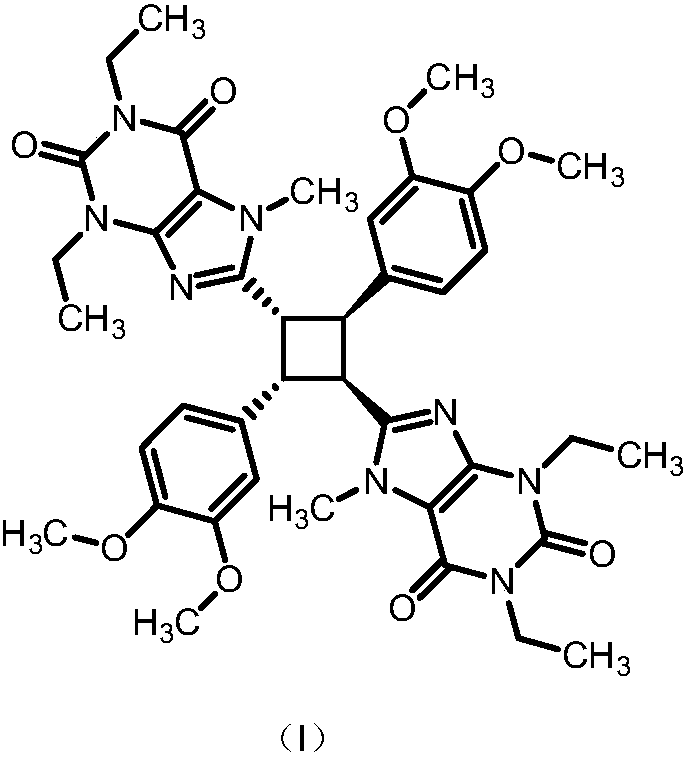
A kind of preparation method of istradefylline impurity standard substance
A technology of istradefylline and standard products, applied in the field of medicine, can solve the problems of cumbersome operation, low yield, large light surface, etc., and achieve an effect that is extremely easy to achieve
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
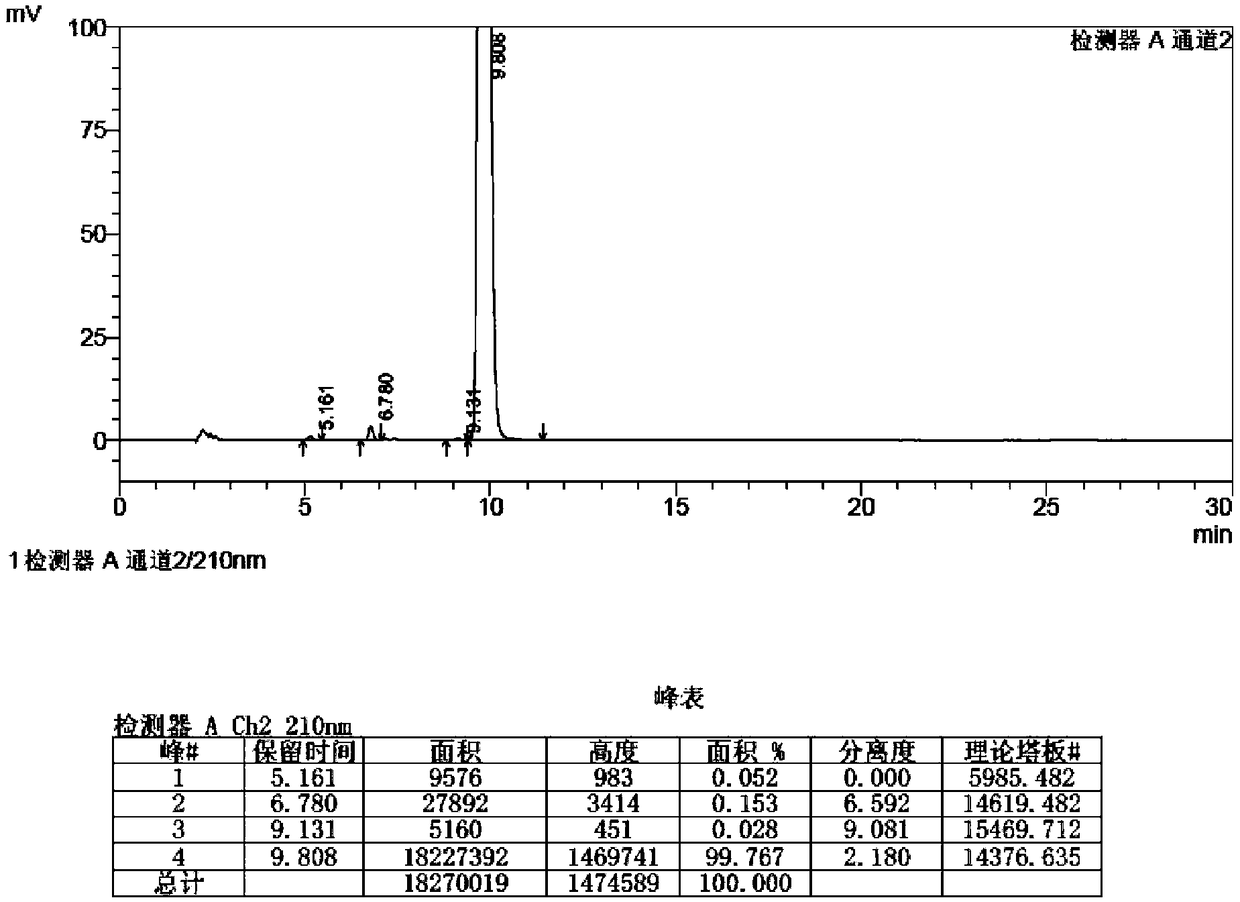
[0040] The preparation method of istradefylline impurity standard substance in the present embodiment is as follows:
[0041] 1. Under the protection of nitrogen, put 200ml of chloroform, 3.5g (26mmol) of aluminum trichloride and 10g (26mmol) of istradefylline into a 500ml colorless glass reaction bottle in sequence, and stir at 30-40°C until completely dissolved , the reaction bottle was tightly closed, placed under a 254nm ultraviolet lamp for 10 hours, and the temperature was controlled at 35-40°C; after the reaction, the reaction solution was washed with 200ml of water and 200ml of saturated aqueous sodium chloride solution, and the organic layer was concentrated to dryness at 30-40°C under reduced pressure. , 9.4 g of impurity crude product was obtained, with a yield of 94.0%.
[0042] 2. Add 100ml of dichloromethane to 9.4g of crude impurities, heat and stir at 30-35°C to dissolve, filter while hot, add 500ml of ethanol to the filtrate, stir and crystallize at 0-10°C for...
Embodiment 2
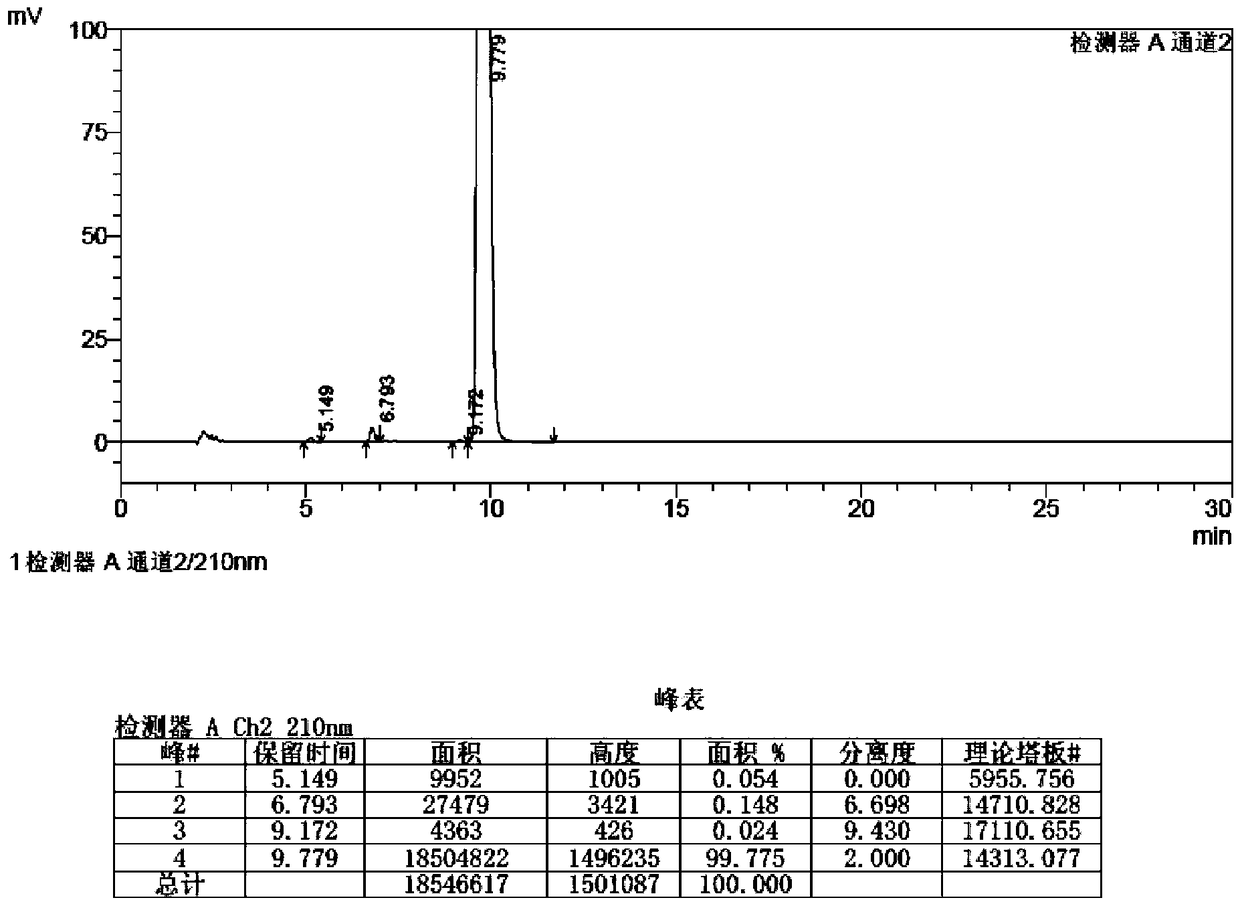
[0053] The preparation method of istradefylline impurity standard substance in the present embodiment is as follows:
[0054] 1. Under the protection of nitrogen, put 500ml of chloroform, 14g (104mmol) of aluminum trichloride and 20g (52mmol) of istradefylline into a 1L colorless glass reaction bottle successively, stir at 30-40°C until completely dissolved, Seal the reaction bottle, place it under a 254nm ultraviolet lamp for 12 hours, and control the temperature at 35-40°C; after the reaction, the reaction solution is washed with 500ml of water and 500ml of saturated aqueous sodium chloride solution, and the organic layer is concentrated to dryness at 30-40°C under reduced pressure. 18.3 g of impurity crude product was obtained with a yield of 91.5%.
[0055] 2. Add 250ml of dichloromethane to 18.3g of crude impurities, heat and stir at 30-35°C to dissolve, filter while hot, add 2000ml of ethanol to the filtrate, stir and crystallize at 0-10°C for 2h, filter, and wash the fi...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More