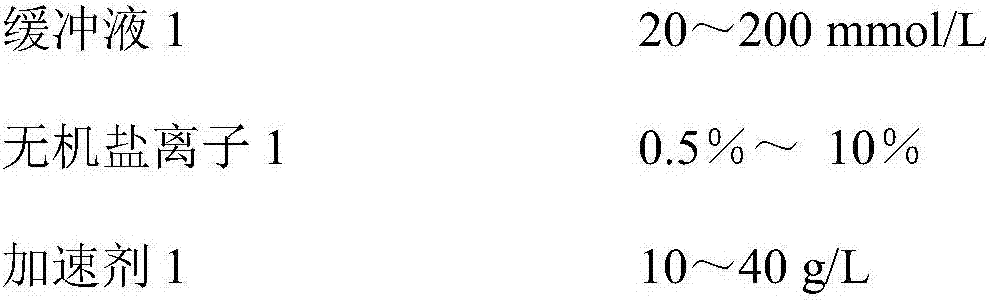Urine microalbumin detection kit and preparation method thereof
A urine microalbumin and detection kit technology, which is applied in the field of medical immunology in vitro diagnosis, can solve the problem of low sensitivity, achieve high sensitivity, simple detection, and improve the effect of detection sensitivity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0046] A urine microalbumin detection kit, comprising reagent R1 and reagent R2 independent of each other, its composition is as follows:
[0047] Reagent R1, its components are:
[0048]
[0049]
[0050] Reagent R2, its component is:
[0051]
Embodiment 2
[0053] A urine microalbumin detection kit, including reagent R1 and reagent R2 independent of each other, its composition is as follows: reagent R1, its components are:
[0054]
[0055] Reagent R2, its component is:
[0056]
[0057]
Embodiment 3
[0059] Preparation and use of the kit:
[0060] 1. Reagent preparation
[0061] A. Preparation of reagent R1:
[0062] Add NaCl 0.5%, PEG6000 25g / L, bovine serum albumin 10g / L, and sodium azide 1g / L to MES buffer 50mmol / L in sequence, stir and mix evenly, and filter with a 0.22 micron mixed cellulose membrane to obtain reagent R1 ;
[0063] B. Preparation of reagent R2:
[0064] Add NaCl 0.5%, sodium azide 1g / L, bovine serum albumin 10g / L, glycerin 30mmol / L, anti-human albumin antibody 3.0g / L to PBS buffer 50mmol / L and stir to mix evenly. The mixed cellulose membrane was filtered to obtain the reagent R2.
[0065] 2. Parameter setting of automatic biochemical analyzer
[0066] a. Detection temperature: 37°C;
[0067] b. Detection wavelength: main wavelength 340nm, secondary wavelength 700nm;
[0068] c. Reaction time: 10 minutes, among them, the incubation time is 5 minutes, read the absorbance A1 immediately after adding the reagent R2, read the absorbance A2 after 5 m...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More