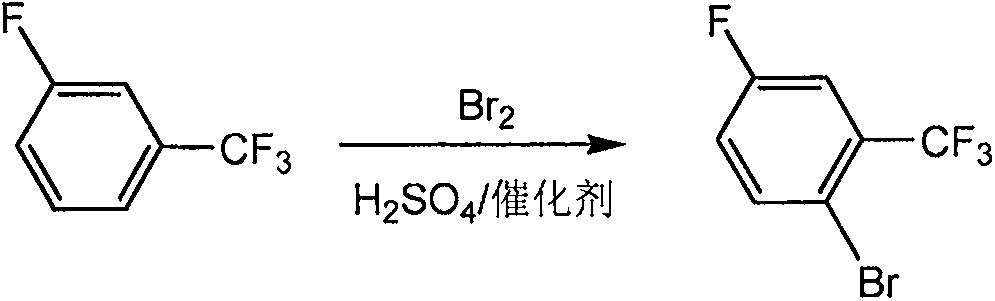
Normal-temperature catalyzed synthesis of 2-bromo-5-fluorobenzotrifluoride
A technology of fluorobenzotrifluoride and normal temperature, which is applied in the field of preparation of chemical intermediate 2-bromo-5-fluorobenzotrifluoride, can solve the problems of environmental hazards, expensive catalysts, high equipment requirements, etc., and achieve significant economic benefits and social benefits. Significant benefits and the effect of reducing production costs
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] In a 1000mL three-necked flask, under normal temperature and pressure conditions, 100g of 100% sulfuric acid, 200g of m-fluorobenzotrifluoride, 4g of ammonium bromide, 2g of bromosuccinimide, and 4g of tetrabutylammonium bromide were successively added under stirring, Add 97.5 g of bromine dropwise. After the addition, control the conditions of normal temperature and pressure, continue the reaction for 5-6 hours, take a sample, and GC detects that the raw material is less than 1%. Terminate the reaction, remove the catalyst by filtration, transfer to a 1000mL separating funnel, stand still for 1h, separate the bottom acid, put the crude product in a 1000mL bottle, add 500g of water under stirring, drop in 5% sodium hydroxide solution to adjust the pH=7, After alkali washing, the solvent was removed, and the fraction at 158-162°C / mmHg was collected by rectification to obtain 278g of the final product, with a yield of 92.3% and a content of 99%.
Embodiment 2
[0031] In a 1000mL three-necked flask, under normal temperature and pressure, add 100% sulfuric acid 180g, m-fluorobenzotrifluoride 200g, ammonium bromide 1g, bromosuccinimide 0.1g, tetrabutylammonium bromide 1g under stirring , add bromine 195g dropwise, after the addition is completed, control the normal temperature and pressure conditions, continue the reaction for 5-6h, take a sample, and GC detects that the raw material is less than 1%. Terminate the reaction, remove the catalyst by filtration, transfer to a 1000mL separating funnel, stand still for 1h, separate the bottom acid, put the crude product in a 1000mL bottle, add 500g of water under stirring, drop in 5% sodium hydroxide solution to adjust the pH=7, After alkali washing, solvent extraction was performed, and the fraction at 158-162° C. / mmHg was collected by rectification to obtain 275 g of the final product, with a yield of 91.2% and a content of 99%.
Embodiment 3
[0033] In a 1000mL three-necked flask, under normal temperature and pressure conditions, 100g of 100% sulfuric acid, 200g of m-fluorobenzotrifluoride, 4.5g of ammonium bromide, 0.5g of bromosuccinimide, and tetrabutylammonium bromide were successively added under stirring. 4.5g, add 97.5g of bromine dropwise, after the addition is completed, control the normal temperature and pressure conditions, continue the reaction for 5-6h, take a sample, and GC detects that the raw material is less than 1%. Terminate the reaction, remove the catalyst by filtration, transfer to a 1000mL separating funnel, stand still for 1h, separate the bottom acid, put the crude product in a 1000mL bottle, add 500g of water under stirring, drop in 5% sodium hydroxide solution to adjust the pH=7, After alkali washing, the solvent was removed, and the fraction at 158-162°C / mmHg was collected by rectification to obtain 276g of the final product, with a yield of 91.5% and a content of 99%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More