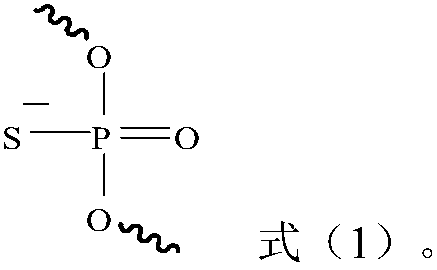Small interfering ribonucleic acid, pharmaceutical composition and application thereof
A nucleotide and nucleotide sequence technology, applied in the field of biomedicine, can solve the problems of poor stability of siRNA, easy to be degraded by nuclease, differences between target nucleic acid species, etc., to inhibit expression, prevent and/or treat blood lipids Abnormal, total cholesterol and triglyceride reduction effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example 1
[0084] The sequence of siRNA is as shown in Table 2, and the positive-sense strand nucleotide sequence numbered as siAN is as shown in SEQ ID NO.4, wherein the 1-19 nucleotide sequence is as shown in SEQ ID NO. The target nucleic acid shown in NO.1 is identical; The antisense strand nucleotide sequence of this siRNA is shown in SEQ ID NO.5, wherein 1-19 nucleotide sequence is shown in SEQ ID NO.1 in table 1 target nucleic acid complementation. The sense strand nucleotide sequence numbered as siAN2 is shown in SEQ ID NO.7, wherein the 1-19 nucleotide sequence is identical to the target nucleic acid shown in SEQ ID NO.6 in the ANGPTL3 mRNA sequence; the antisense of the siRNA The chain nucleotide sequence is shown as SEQ ID NO.8, wherein the 1-19 nucleotide sequence is complementary to the target nucleic acid shown as SEQ ID NO.6 in Table 1.
[0085] As shown in Table 2, in this preparation example, an siRNA with the nucleotide sequence of the sense strand as shown in SEQ ID NO...
Embodiment 1
[0094]This example is used to detect the inhibitory rate of siRNA obtained in Preparation Example 1 on the expression level of ANGPTL3 mRNA in vitro.
[0095] The human liver cancer cell line Huh7 was inoculated in 24-well plates with DMEM complete medium containing 10% fetal bovine serum at a seeding density of 4×10 5 Cells / well, 0.5mL culture medium per well, culture overnight at 37°C.
[0096] Aspirate the cell culture medium in the 24-well plate, and add 0.5 mL Opti-MEM serum-free medium to each well. Dilute 1.5 μL of the siRNA in Preparation Example 1 and Preparation Example 2 at a concentration of 20 μM with 50 μL Opti-MEM serum-free medium; add 1 μL Lipofectamine TM 2000 (Invitrogen Company) was diluted in 50 μL Opti-MEM serum-free medium, incubated at room temperature for 5 minutes after mixing; mixed diluted siRNA and diluted Lipofectamine TM 2000, mix gently, and let stand at room temperature for 20 minutes to allow complex formation. Add the above final mixed s...
preparation example 2
[0105] The siRNA obtained after chemical modification of the siRNA sense strand and antisense strand numbered siNC is shown in Table 5, and the number is siNC-M; the four groups of siRNA obtained after chemical modification of the siRNA sense strand and antisense strand numbered siAN are shown in Table 5. 5, respectively numbered siAN-M1, siAN-M2, siAN-M3, siAN-M4. Wherein (M) represents that the pentose group in the nucleotide residue on its left side is 2'-methoxyribose group, (F) represents that the pentose group in the nucleotide residue on its left side is 2' - Fluorinated ribose; S represents that the phosphate group between the deoxyribonucleotide residues dTdT on the left and right sides is a phosphorothioate group.
[0106] table 5
[0107]
[0108]
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
| diameter | aaaaa | aaaaa |
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More