Poly (HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent and preparation method thereof
One-DOTA, nuclear magnetic resonance technology, applied in the direction of nuclear magnetic resonance/magnetic resonance imaging contrast agents, pharmaceutical formulations, preparations for in vivo tests, etc., can solve problems such as large dosage, short imaging window time, and low relaxation efficiency , to achieve the effect of increasing imaging effect, increasing passive targeting ability and prolonging cycle time
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
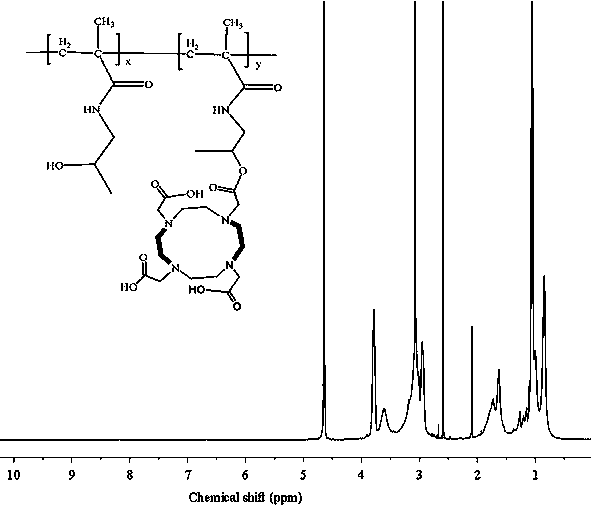
[0039] Example 1 Poly(HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent, the structural formula of the contrast agent is as follows:
[0040] ;
[0041] Where: x = 90 mol%, y = 10 mol%.
[0042] The average molecular weight of the contrast agent is 20-40 kDa.
[0043] The preparation method of this poly(HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent comprises the following steps:
[0044] (1) Preparation of poly(HPMA) polymer:
[0045] Will N-(2-Hydroxypropyl)methacrylamide (0.14g, 1.00 mmol) was first dissolved in 0.5 mL of DMSO at 25°C, then 0.5 mL of acetone was added, stirred until dissolved, and 0.065 g ( 5%, wt) initiator azobisisobutyronitrile (AIBN), vacuumize and inflate with nitrogen for 3 to 5 times, seal and react at 50°C for 24 h under nitrogen protection, precipitate and filter with a mixture of acetone and ether, Precipitate A was obtained, which was dissolved in 1 mL of anhydrous methanol, centrifuged in an ultrafiltration centrifuge tube ...
Embodiment 2
[0060] Example 2 Poly(HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent, the structural formula of the contrast agent is as follows:
[0061] ;
[0062] Where: x = 80 mol%, y = 20 mol%.
[0063] The average molecular weight of the contrast agent is 20-40kDa.
[0064] The preparation method of this poly(HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent comprises the following steps:
[0065] (1) Preparation of poly(HPMA) polymer:
[0066] Will N -(2-Hydroxypropyl)methacrylamide (0.2 g, 1.4 mmol) was first dissolved in 1.0 mL of DMSO at 25°C, then 3.0 mL of acetone was added, stirred until dissolved, and 0.016 g ( 8%, wt) initiator azobisisobutyronitrile (AIBN), vacuumize and inflate with nitrogen for 3 to 5 times, seal and react at 55°C for 22 h under nitrogen protection, precipitate and filter through a mixture of acetone and ether, The precipitate A was obtained. After the precipitate A was dissolved in 1.0 mL of anhydrous methanol, it was centrifuged in an...
Embodiment 3
[0075] Example 3 Poly(HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent, the structural formula of the contrast agent is as follows:
[0076] ;
[0077] Where: x = 10 mol%, y = 90 mol%.
[0078] The average molecular weight of the contrast agent is 20-40kDa.
[0079] The preparation method of this poly(HPMA)-DOTA-Gd nuclear magnetic resonance contrast agent comprises the following steps:
[0080] (1) Preparation of poly(HPMA) polymer:
[0081] Will N -(2-Hydroxypropyl)methacrylamide (0.4 g, 2.8 mmol) was first dissolved in 1.0 mL of DMSO at 25 °C, then 3 mL of acetone was added, stirred until dissolved, and 0.040 g ( 10 %, wt) initiator azobisisobutyronitrile (AIBN), vacuumize and inflate nitrogen for 3 to 5 times, seal and react at 60°C for 20 h under nitrogen protection, precipitate and filter through a mixture of acetone and ether, The precipitate A was obtained. After the precipitate A was dissolved in 1.0 mL of anhydrous methanol, it was centrifuged through an ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Average molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More