Antibody composition for recognizing cancer stem cell components in gastric poorly differentiated adenocarcinoma and application thereof
A technology of gastric poorly differentiated adenocarcinoma and antibody composition, which is applied in the field of biological detection, can solve the problems of lack of specific pathological diagnostic markers of cancer stem cell components, and does not consider the difference in prognosis of gastric cancer stem cell components, so as to shorten the reporting period and reduce the interpretation time. Difficulty, the effect of simplifying the operation process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
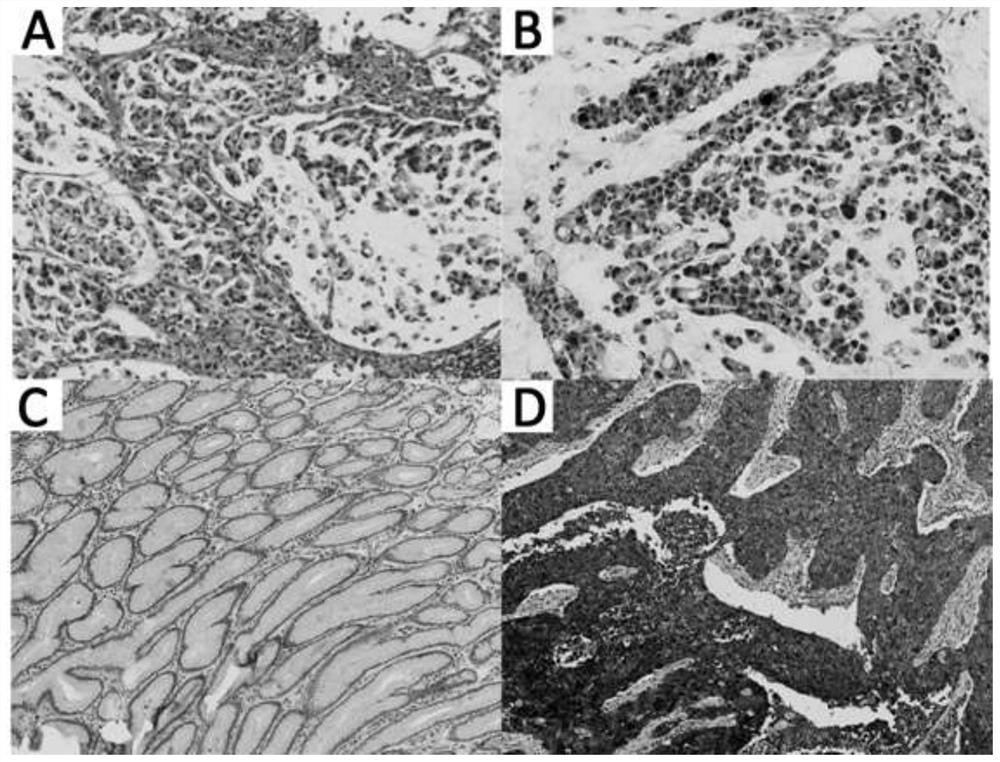
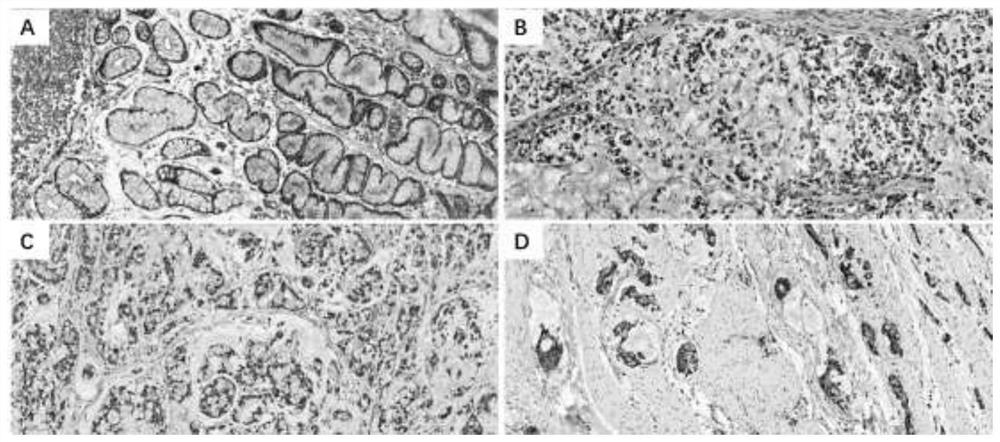
[0027] Paraffin section specimens: from 38 patients with gastric diseases hospitalized in the First Affiliated Hospital of Nanjing Medical University from January 2010 to June 2019. All patients were confirmed by pathological diagnosis. The basic information of the patients is shown in Table 1. The median age was 64.5 years, including 26 male patients and 12 female patients. Among them, there were 18 cases of gastritis, 18 cases of poorly differentiated gastric adenocarcinoma, and 2 cases of primary gastric adenosquamous cell carcinoma.
[0028] Table 1. Basic information of 38 patients with confirmed gastric diseases
[0029]
[0030]
[0031] Paraffin section immunohistochemistry: Select 4 μm paraffin-embedded human gastric tissue sections and incubate at 60°C for 1 hour. Soak in xylene for 30 minutes to dewax, and then soak in gradient ethanol solution in turn. Block with 3% hydrogen peroxide solution in methanol for 10 minutes at room temperature. Wash with PBS 3 ...
Embodiment 2
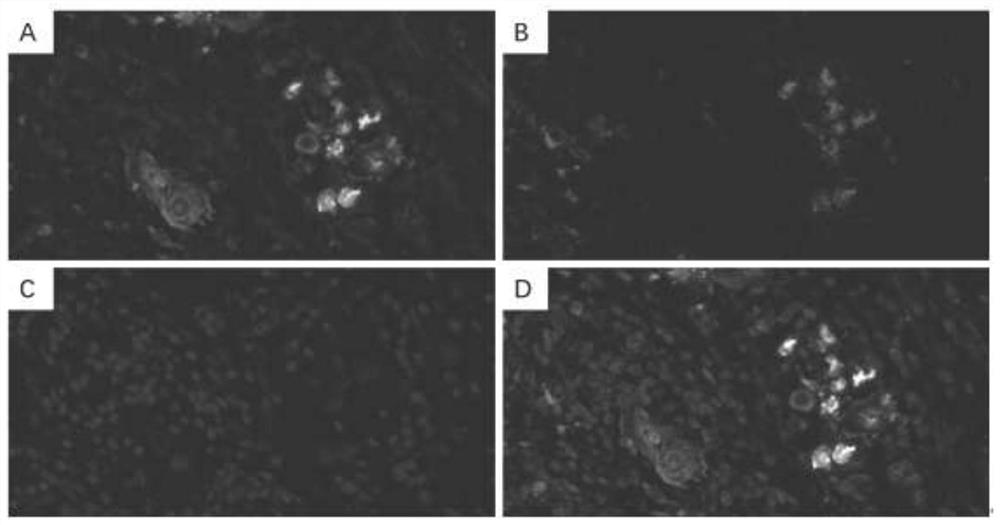
[0036] Four of the 18 poorly differentiated gastric adenocarcinomas with positive expression of SCCA were selected, and their paraffin sections were immunofluorescence stained for SCCA and ALDH1.
[0037] Immunofluorescent staining of paraffin sections: select 4 μm paraffin-embedded human gastric tissue sections and incubate at 60°C for 1 hour. Soak in xylene for 30 minutes to dewax, and then soak in gradient ethanol solution in turn. A part of the section was stained with HE for histomorphological analysis; the other part was immunofluorescent stained, and the antigen was retrieved in citrate buffer (pH 6.0), blocked in PBS containing 5% goat serum, and incubated with the primary antibody at 4°C overnight. Wash with PBS 3 times, 5 minutes each time. Fluorescently labeled goat anti-mouse secondary antibody and goat anti-rabbit secondary antibody were incubated at room temperature for 2 hours. Wash with PBS 3 times, 5 minutes each time. Add anti-fluorescence quenching reage...
Embodiment 3
[0042] Isolate gastric tissue:
[0043] 1) Put the freshly resected gastric tissue pathologically confirmed as poorly differentiated adenocarcinoma into pre-cooled DMEM / F12 medium containing 10% fetal bovine serum;
[0044] 2) Rinse the tissue block with pre-cooled sterile PBS in the ultra-clean bench, use sterile ophthalmic scissors to break the tissue block to about 1 mm, and transfer it to a digestion bottle;
[0045] 3) Configure DMEM / F12 medium containing type IV collagenase, type II collagenase and hyaluronidase, add it to the digestion bottle, ensure that the tissue block is completely submerged and suspended, and seal it with sterile tinfoil;
[0046] 4) Digest with horizontal shaking at 37°C until the tissue blocks disappear;
[0047] 5) After digestion, the cell suspension was filtered through a 200-mesh sieve, and the obtained supernatant was centrifuged at 1000 rpm / min for 5 minutes;
[0048] 6) Discard the supernatant, resuspend in pre-cooled PBS, and centrifuge...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap