Natural Chinese herbal medicine plant feed additive and preparation technology thereof
A technology of natural Chinese herbal medicine and plant feed, which is applied in the field of natural Chinese herbal medicine plant feed additive and its preparation process, can solve the problems of lack of high efficiency and low toxicity, and achieve the effect of rich raw materials and simple production process
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
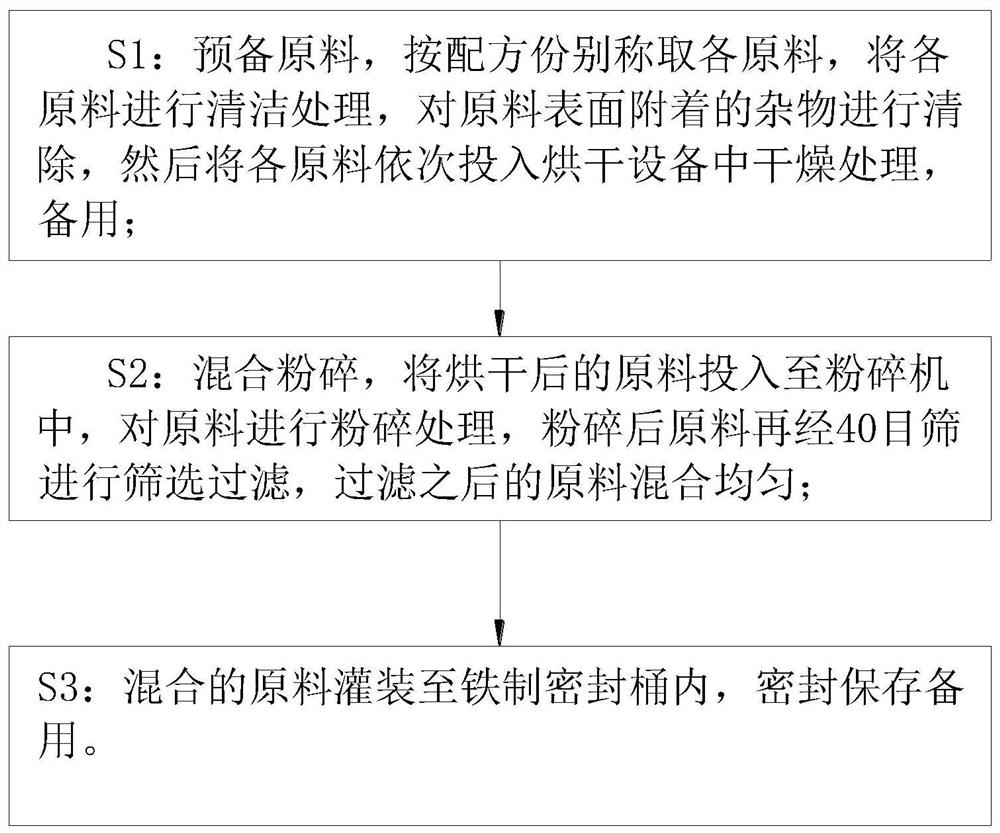
[0023] A natural Chinese herbal medicine plant feed additive, which contains: 4 parts of tangerine peel, 3 parts of Shouwu, 5 parts of Qinpi, 3 parts of astragalus, 2 parts of Codonopsis pilosula, 4 parts of Perilla, 3 parts of Guanzhong and 5 parts of Artemisia argyi , 2 parts of Ganoderma lucidum, 4 parts of Dodder, 4 parts of Eucommia, and 3 parts of Psoralen, the above-mentioned raw materials are processed and produced according to their preparation process, and the additives are sealed and preserved for subsequent use after production;
Embodiment 2
[0025] A natural Chinese herbal medicine plant feed additive, which contains: 5 parts of tangerine peel, 4 parts of Shouwu, 6 parts of qinpi, 4 parts of astragalus, 3 parts of Codonopsis pilosula, 5 parts of perilla, 4 parts of perilla, and 6 parts of mugwort leaves according to the ratio of parts by mass , 3 parts of ganoderma lucidum, 5 parts of dodder seed, 5 parts of Eucommia ulmoides, 4 parts of psoralen, the above-mentioned raw materials are processed and produced according to its preparation process, and the additives are sealed and preserved for subsequent use after production;
Embodiment 3
[0027] A natural Chinese herbal medicine plant feed additive, which contains: 6 parts of tangerine peel, 5 parts of Shouwu, 7 parts of Qinpi, 5 parts of astragalus, 4 parts of Codonopsis pilosula, 6 parts of perilla, 5 parts of perilla, and 7 parts of Artemisia argyi , 4 parts of Ganoderma lucidum, 6 parts of Dodder, 6 parts of Eucommia, and 5 parts of Psoralen, the above-mentioned raw materials are processed and produced according to its preparation process, and the additives are sealed and preserved for subsequent use after production;
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More