An anti-new coronavirus fully human broad-spectrum neutralizing antibody zwc12 and its application
A fully human, monoclonal antibody technology used in the fields of microbiology and immunology to solve problems such as loss of neutralizing activity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0036] Example 1 Screening and preparation of human anti-SARS-CoV-2 monoclonal antibodies.
[0037] 1. Collection of blood samples.
[0038] After obtaining informed consent, 20 mL of blood samples were collected from the recipients immunized with the recombinant novel coronavirus vaccine 14 days after the second immunization for subsequent experiments.
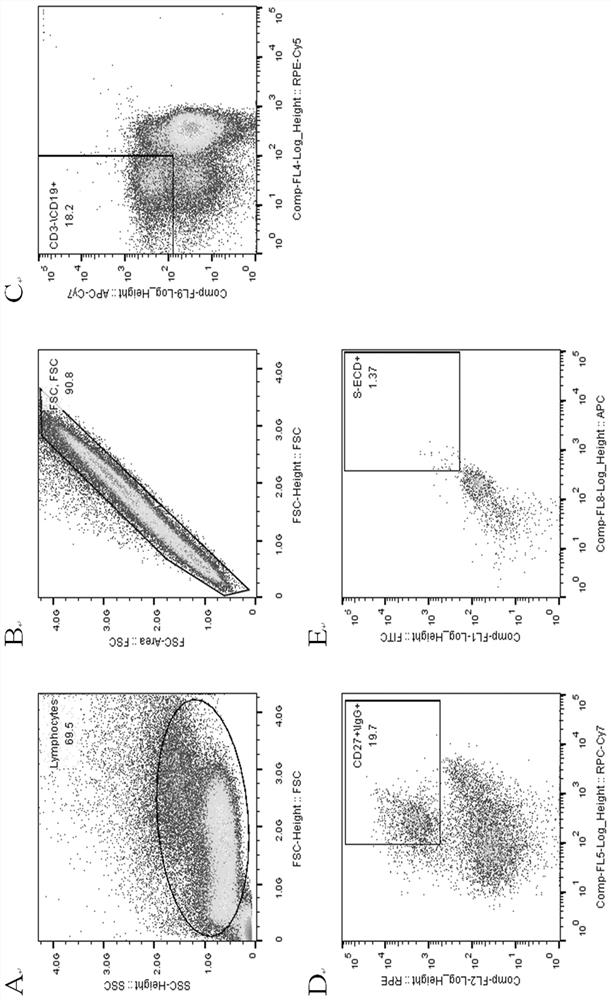
[0039] 2. Sorting memory B cells by flow cytometry.
[0040] The collected blood samples were separated from PBMC by Ficoll density gradient centrifugation, and the process was as follows:
[0041] 1) Take fresh anticoagulated whole blood and anticoagulate with EDTA.
[0042] 2) Add the same volume of separation liquid as the blood sample into the centrifuge tube, spread the blood sample above the liquid surface of the separation liquid, and keep the interface between the two liquid surfaces clear.
[0043] 3) Trim, room temperature, horizontal rotor 800 g, acceleration and deceleration speed 3, centrifuge for 30 min.
[...
Embodiment 2
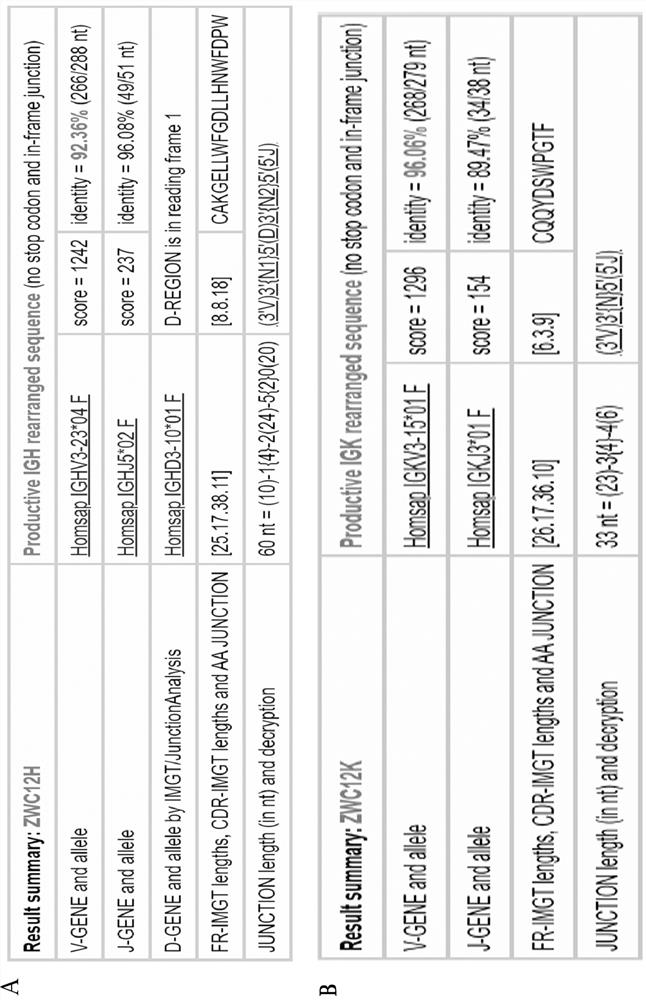
[0119] Example 2. Analysis of the epitope recognized by antibody ZWC12.
[0120] 1) Coating: Take the recombinant SARS-CoV-2 WT (new crown wild strain Genbank number: NC_045512.2) S-ECD antigen, S1 antigen, RBD antigen and S2 antigen on the 96-well enzyme-linked plate the day before the experiment. solution diluted to a concentration of 2 μg / mL, coated with 100 μL per well, and coated overnight at 4°C.
[0121] 2) Blocking: Wash 3 times with a plate washer (BIO-TEK, 405_LS) on the day of the experiment, add 100 µL of blocking solution to each well, and incubate at 37°C for 1 hour.
[0122] 3) Sample incubation: wash the plate 3 times, add 100 μL diluent to each well except the first well, dilute the antibody to 1 μg / mL in the first well, 4-fold serial dilution, 100 μL / well, set three for each antibody Duplicate the wells and incubate at 37°C for 1 h.
[0123] 4) Secondary antibody incubation: wash the plate 3 times, dilute the HPR-labeled goat anti-human IgG secondary antibo...
Embodiment 3
[0129] Example 3: Identification of cross-binding activity of antibody ZWC12.
[0130] The cross-binding activity between ZWC12 and the S protein of SARS-CoV-2 variants of concern (Variants of concern) was identified, the method is the same as above, and the results are as follows Figure 7 shown. ZWC12 specifically binds to the S-ECD protein of Alpha strain, Beta strain, Gamma strain, Delta strain, and Omicron strain, and exhibits a dose-response relationship. The results showed that monoclonal antibody ZWC12 could cross-link the S-ECD protein of Alpha strain, Beta strain, Gamma strain, Delta strain and Omicron strain.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More