Alternative Compositions and Methods for the Culture of Stem Cells
a technology of stem cell culture and composition, which is applied in the field of stem cell culture, can solve the problems of inability to maintain human es cells in a pluripotent state for long or indefinite periods, and lack of evidence in the prior art for long-term isolation and/or maintenance of human pluripotent es cells on non-fibroblast feeder cells,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Isolation of a Human Pluripotent ES Cell on a Mouse Embryonic Fibroblast Feeder Cell Layer
[0103]Embryo Collection: Human embryos were obtained as described by Thomson et al., 1998, supra. Briefly, fresh or frozen cleavage stage human embryos were produced by in vitro fertilization (IVF) for clinical purposes. The embryos were donated by individuals after informed consent and after institutional review board approval. The donated human embryos were cultured to the blastocyst stage in IVC-1 Medium (In Vitro Care, San Diego, Calif.) from day 0 until day 3 or day 4. After 3 days of culture the embryos were visually assessed by qualified embryologists for their quality and likely ability to develop normally. Embryos of high quality were transferred either to the patients from whom the ova were collected or were donated to other patients. The remaining embryos were cultured in IVC-3 blastocyst medium (In Vitro Care, San Diego, Calif.) and reassessed at 5 days in vitro. High quality embryo...
example 2
Characterization of a Human Pluripotent ES Cell Isolated on MEFs
[0110]The human ES cell lines that were generated in Example 1 were characterized to ensure that they were pluripotent.
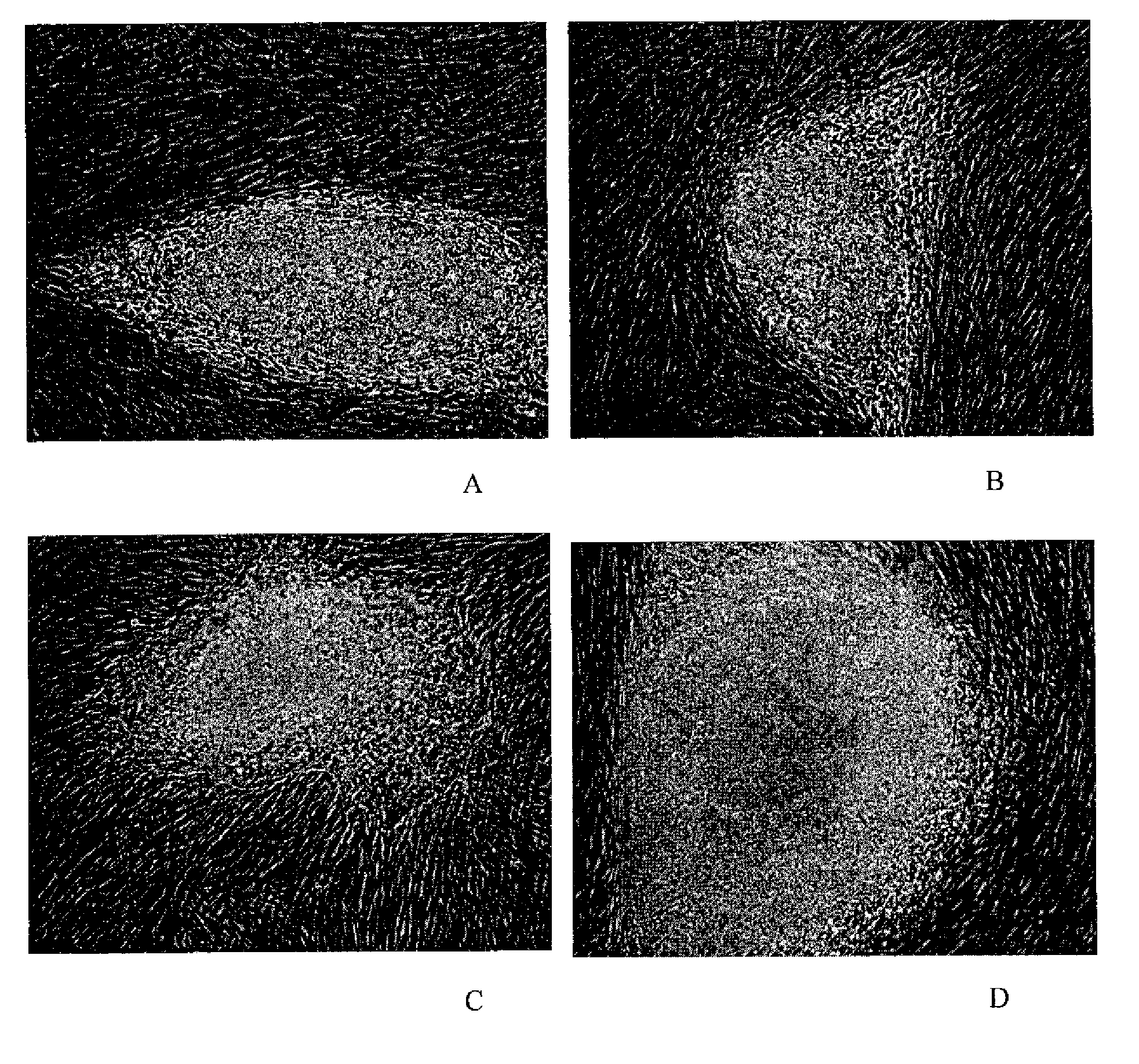
[0111]Morphology: The morphology of the derived human ES cell was similar in at least some ways to the human ES cell lines derived by other investigators (Thomson et al., 1998 supra; Reubinoff et al., 2000 supra). The human ES cells have a high nucleus / cytoplasm ratio and prominent nucleoli. Additionally, the borders between the individual ES cells are very distinguishable, particularly in comparison to mouse ES cells. The characteristic morphology of human ES cells allows for the ready identification of the cells.
[0112]Gene expression studies: The human ES cell lines generated in Example 1 expressed alkaline phosphatase and Oct-4, two molecular markers of pluripotent cells.
[0113]Alkaline phosphatase was visualized using the Vector Red alkaline phosphatase substrate kit (Vector Lab, Burlingame, Calif.)....
example 3
Maintenance of a Human Pluripotent ES Cell on a Human Granulosa Feeder Cell Layer
[0119]Isolation of granulosa cells: Human granulosa cells were harvested at the time oocytes were collected for IVF. The oocytes were collected by laporoscopy. Oocytes have a tightly associated layer of cells surrounding them, known as the cumulus oophorus. The granulosa cells that comprise the cumulus oophorus were stripped from the oocytes by hyaluronidase treatment. The hyaluronidase treatment was followed by repeated passage of the granulosa cells through a narrow bore pipette. The granulosa cells were isolated as clumps of cells, which were further broken down to a single cell suspension by a 2 minute trypsin-EDTA treatment with 0.25% trypsin-EDTA in PBS (Gibco-BRL). After the trypsin treatment, the granulosa cells were washed with HES cell medium and plated on tissue culture dishes. The granulosa cells were cultured at 37° C. and 5% CO2.
[0120]The granulosa cells were cultured in HES medium. After ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More