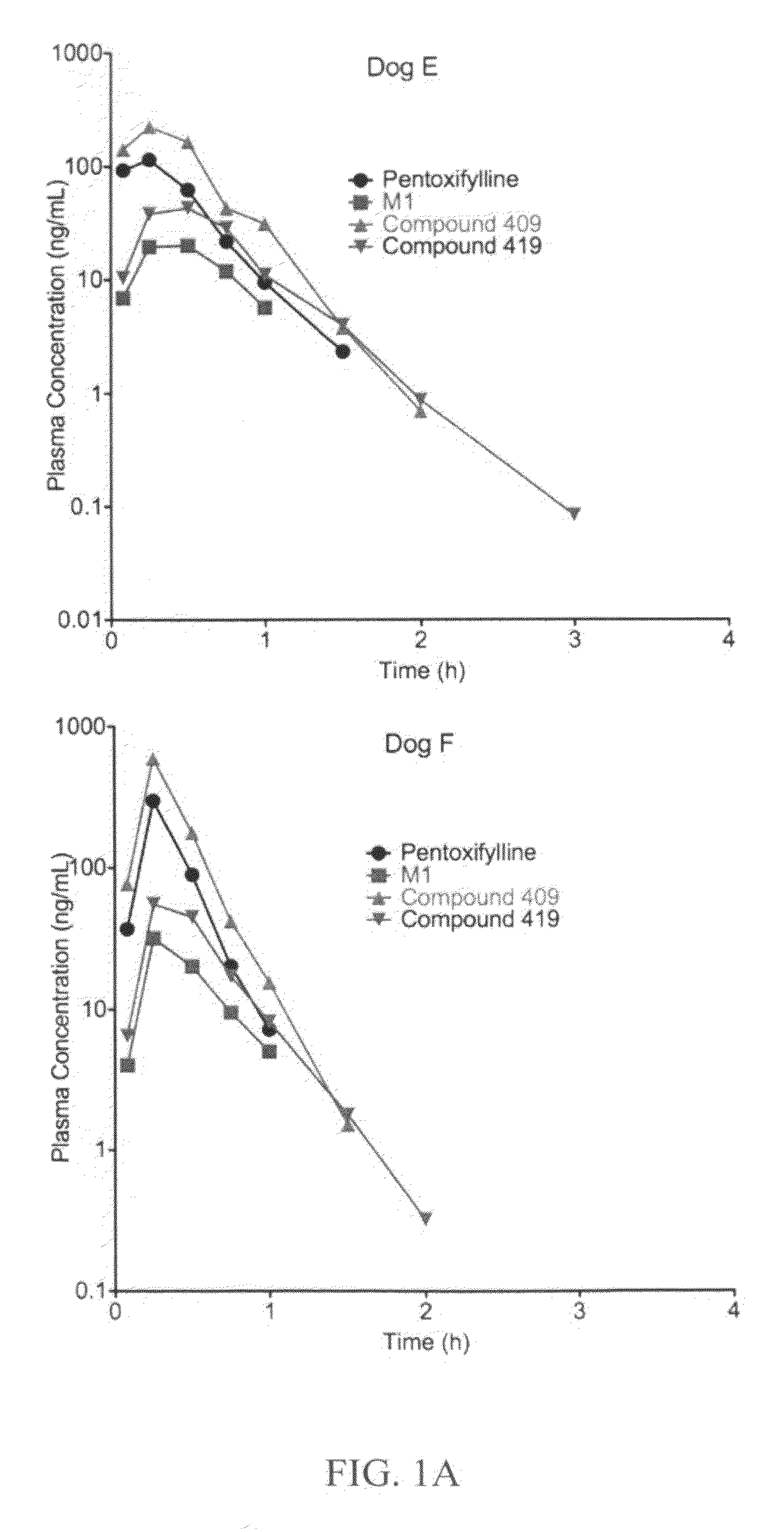
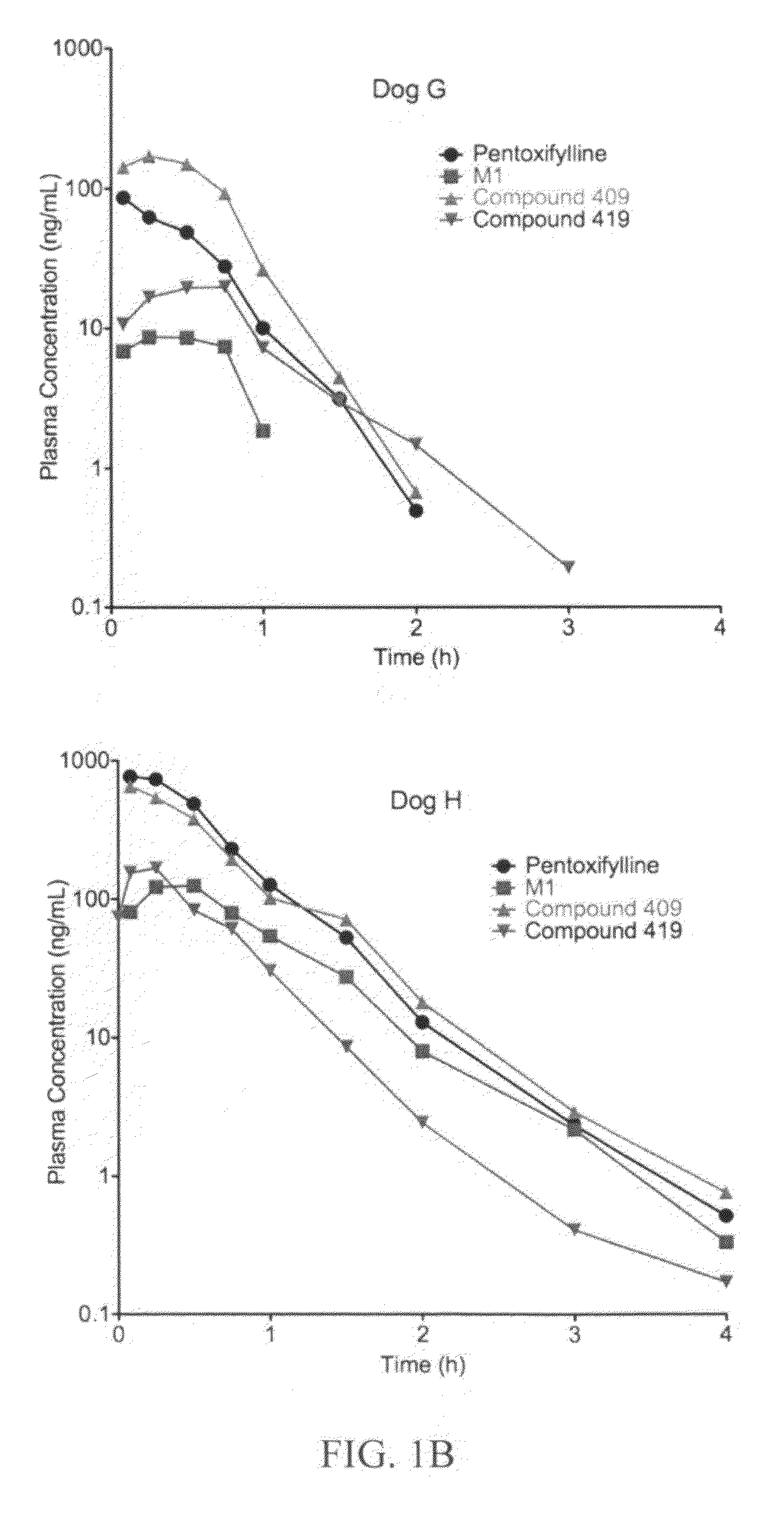
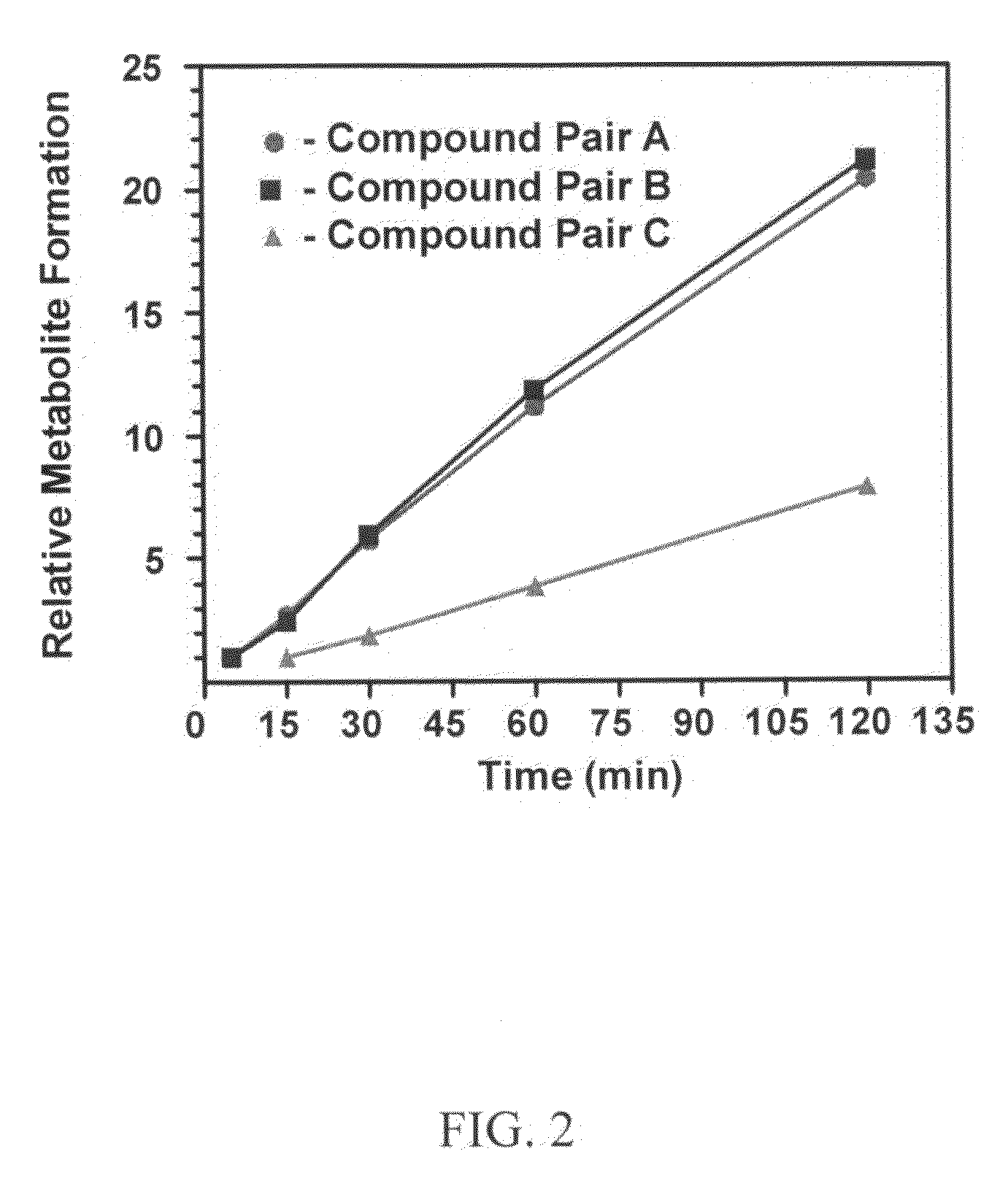
Substituted xanthine derivatives
a technology of substituted xanthine and derivatives, which is applied in the field of substituted xanthine derivatives, can solve the problem that the risk of toxic reactions to pentoxifylline may be greater in patients suffering from renal impairmen
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of 3-Methyl-7-(methyl-d3)-1-(5-oxohexyl)-1H-purine-2,6(3H,7H)-dione (Compound 100)
[0217]
[0218]Step 1. 3-Methyl-7-(methyl-d3)-1H-purine-2,6(3H,7H)-dione (51). A suspension of 3-methylxanthine 50 (5.0 g, 30.1 mmol, 1 equiv) and powdered K2CO3 (5.0 g, 36.0 mmol, 1.2 equiv) in DMF (95 mL) was heated to 60° C. and iodomethane-d3 (Cambridge Isotopes, 99.5 atom % D, 2.2 mL, 36.0 mmol, 1.2 equiv) was added via syringe. The resulting mixture was heated at 80° C. for 5 hours (h). The reaction mixture was cooled to room temperature (rt) and the DMF was evaporated under reduced pressure. The crude residue was dissolved in 5% aqueous NaOH (50 mL), resulting in a dull yellow solution. The aqueous solution was washed with DCM three times (500 mL total). The aqueous layer was acidified to pH 5 with acetic acid (6 mL), resulting in formation of a tan precipitate. The mixture was cooled in an ice-water bath, and the solids were filtered and washed with cold water. The solid was dried in a v...
example 2
Synthesis of 8-d1-3-methyl-7-(methyl-d3)-1-(6-d3-4-d2-5-oxohexyl)-1H-purine-2,6(3H,7H)-dione (Compound 409)
[0221]8-d1-3-methyl-7-(methyl-d3)-1-(6-d3-4-d2-5-oxohexyl)-1H-purine-2,6(3H,7H)-dione (Compound 409). A suspension of 100 (1.80 g, 6.4 mmol, 1 equiv) and powdered K2CO3 (0.23 g, 1.7 mmol, 0.25 equiv) in D2O (Cambridge Isotope Labs, 99 atom % D) (45 mL) was stirred under reflux conditions for 24 h during which time the suspension became a slightly yellow solution. The reaction mixture was cooled to rt, saturated with sodium chloride, and extracted four times with dichloromethane (400 mL total). The combined organic solution was dried over Na2SO4, filtered, and evaporated under reduced pressure to provide 1.7 g of a slightly yellow oil that solidified upon standing. The crude material was re-subjected to the hydrogen / deuterium exchange conditions described above with fresh K2CO3 and D2O. After an identical workup, the off-white solid was triturated with hexanes (100 mL) and filte...
example 3
Synthesis of 3,7-Di(methyl-d3)-1-(5-oxohexyl)-1H-purine-2,6(3H,7H)-dione (Compound 101)
[0223]
[0224]Step 1. 3,7-Di(methyl-d3)-1H-purine-2,6(3H,7H)-dione (55). A suspension of xanthine 53 (2.00 g, 13.2 mmol, 1.0 equiv) and hexamethyldisilazane (32 mL) in toluene (60 mL) was heated to reflux and stirred for 4 days. The reaction mixture was cooled to room temperature, diluted with additional toluene (50 mL) and filtered through Celite to remove any unreacted starting material. The filtrate was evaporated to dryness under reduced pressure to produce 54 as a white solid (4.1 g). A portion of this material (3.00 g) was placed in a 100 mL sealed tube reaction vessel, followed by the addition of toluene (60 mL) and CD3I (4 mL, Cambridge Isotopes, 99.5 atom % D). The reaction mixture was heated in a 120° C. oil bath and stirred for 24 hours, during which time the reaction mixture turned yellow and a solid formed. The reaction mixture was cooled to room temperature, resulting in the entire rea...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More