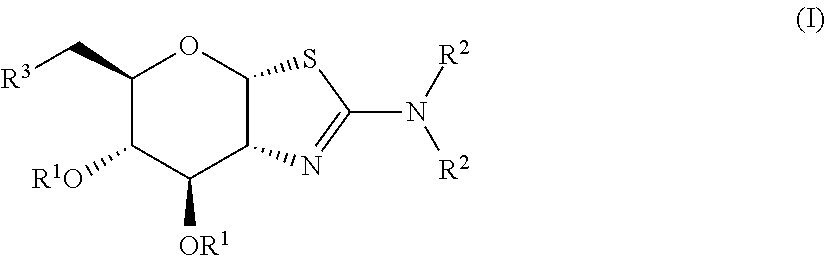
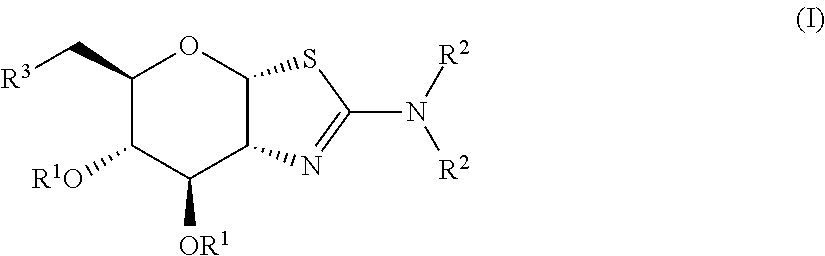
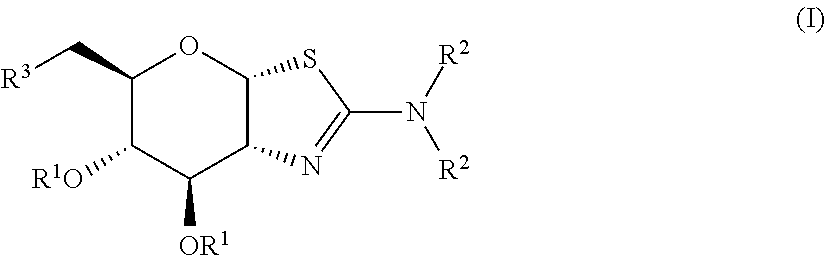
Selective glycosidase inhibitors and uses thereof
a glycosidase inhibitor and selective technology, applied in the field of selective glycosidase inhibitors, can solve the problems of complex use of non-selective inhibitors in studying the physiological role of one particular enzyme, hyperphosphorylation of tau, and disruption of normal functions of tau, so as to increase the level of o-glcnac
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
[0144]The following examples are intended to illustrate embodiments of the invention and are not intended to be construed in a limiting manner.
examples 1 & 2
(3aR,5R,6S,7R,7aR)-5-(acetoxymethyl)-2-(azetidin-1-yl)-5,6,7,7a-tetrahydro-3aH-pyrano[3,2-d]thiazole-6,7-diyl diacetate (2) and (3aR,5R,6S,7R,7aR)-2-(azetidin-1-yl)-5-(hydroxymethyl)-5,6,7,7a-tetrahydro-3aH-pyrano[3,2-d]thiazole-6,7-diol (3)
[0145]
(3aR,5R,6S,7R,7aR)-5-(acetoxymethyl)-2-(azetidin-1-yl)-5,6,7,7a-tetrahydro-3aH-pyrano[3,2-d]thiazole-6,7-diyl diacetate (2)
[0146]A solution of azetidine hydrochloride (12 g, 129 mmol) and (3R,4R,5S,6R)-6-(acetoxymethyl)-3-isothiocyanato-tetrahydro-2H-pyran-2,4,5-triyl triacetate (48 g, 123 mmol) in dichloromethane (500 mL) was treated with triethylamine (18.7 g, 185 mmol) for 1 h at room temperature, and followed by addition oftrifluoroacetic acid (56.2 g, 493 mmol). The reaction mixture was stirred overnight at room temperature, and then quenched by aqueous sodium bicarbonate. The organic layer was separated, dried over anhydrous MgSO4, and condensed under vacuum to give a residue, which was purified by a silica gel column, eluted with 1% ...
example 15
(3aR,5R,6S,7R,7aR)-2-(azetidin-1-yl)-5-((cyclopentylamino)methyl)-5,6,7,7a-tetrahydro-3aH-pyrano[3,2-d]thiazole-6,7-diol (5)
[0150]
((3aR,5R,6S,7R,7aR)-2-(azetidin-1-yl)-6,7-dihydroxy-5,6,7,7a-tetrahydro-3aH-pyrano[3,2-d]thiazol-5-yl)methyl 4-methylbenzenesulfonate (4)
[0151]A solution of (3aR,5R,6S,7R,7aR)-2-(azetidin-1-yl)-5-(hydroxymethyl)-5,6,7,7a-tetrahydro-3aH-pyrano[3,2-d]thiazole-6,7-diol (5.0 g, 19 mmol) and triethylamine (3.88 g, 38 mmol) in DMF (50 mL) was treated with 4-methylbenzene-1-sulfonyl chloride (4.4 g, 23 mmol) overnight at room temperature. The reaction mixture was quenched by water (100 mL), extracted with dichloromethane (3×80 mL), washed with brine (3×50 mL), dried over anhydrous magnesium sulfate, and concentrated under vacuum to give a residue, which was purified by a silica gel column eluted with 1%-5% methanol in dichloromethane to give crude compound 4 as a light yellow solid (1.0 g). This material was employed in the next step without further purification...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Flow rate | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| concentrations | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More