Treatment of cancer using Anti-cd19 chimeric antigen receptor
a technology of chimeric antigen receptor and cancer, which is applied in the direction of immunoglobulins, peptides, drugs against animals/humans, etc., can solve the problems of difficult to achieve clinical effectiveness, difficult to improve immunogenicity, and serious side effects of traditional treatment options, so as to improve the efficacy of cells expressing car molecule and improve the effect of one or more side effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
ion of Murine Anti-CD19 Antibody
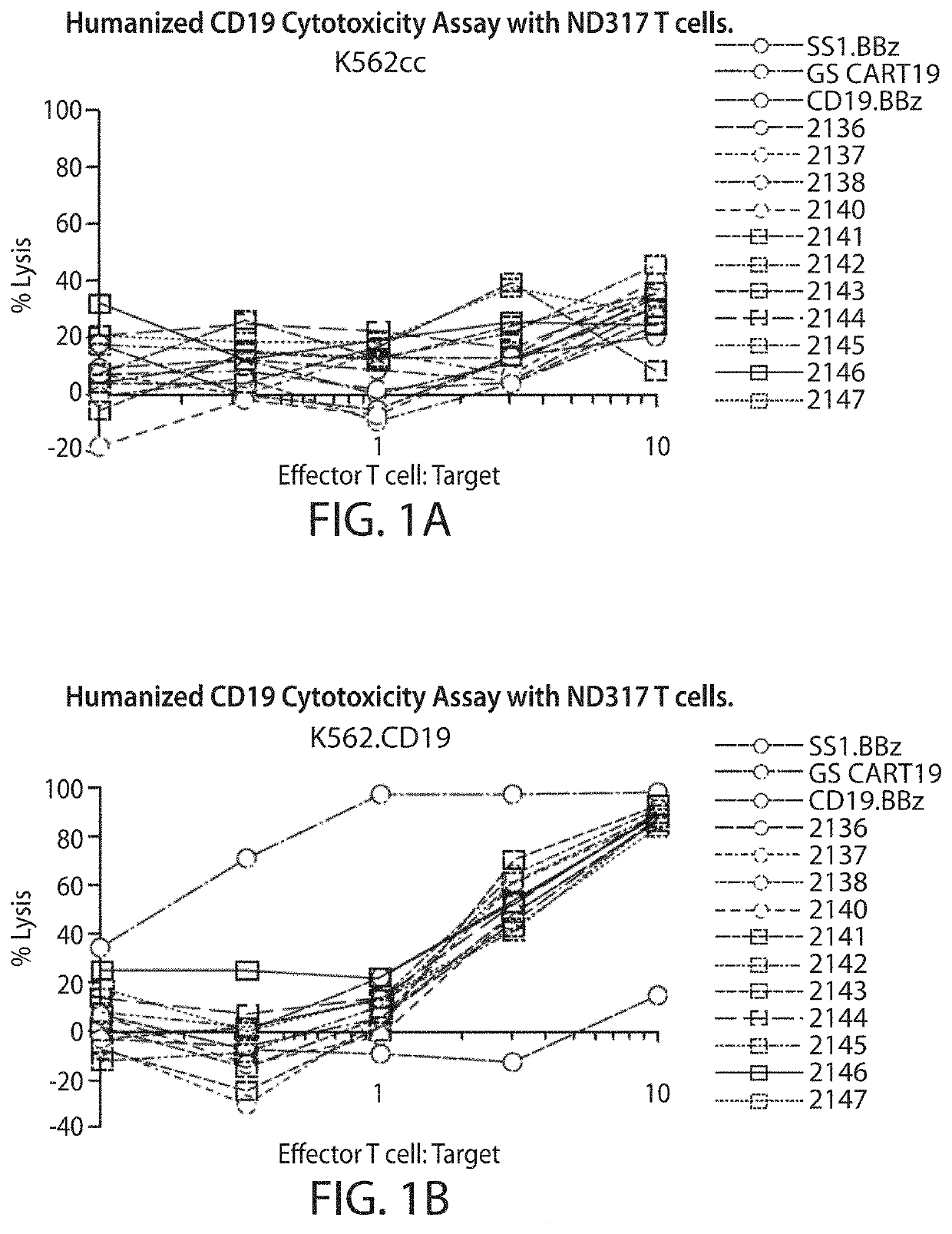
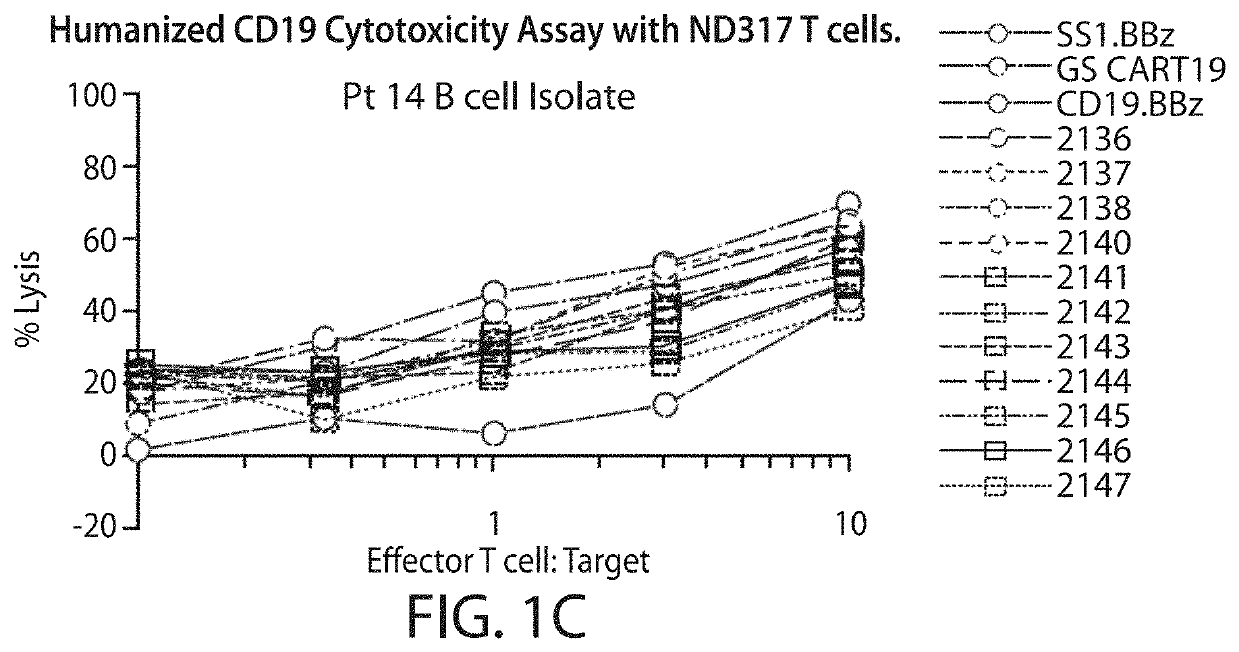
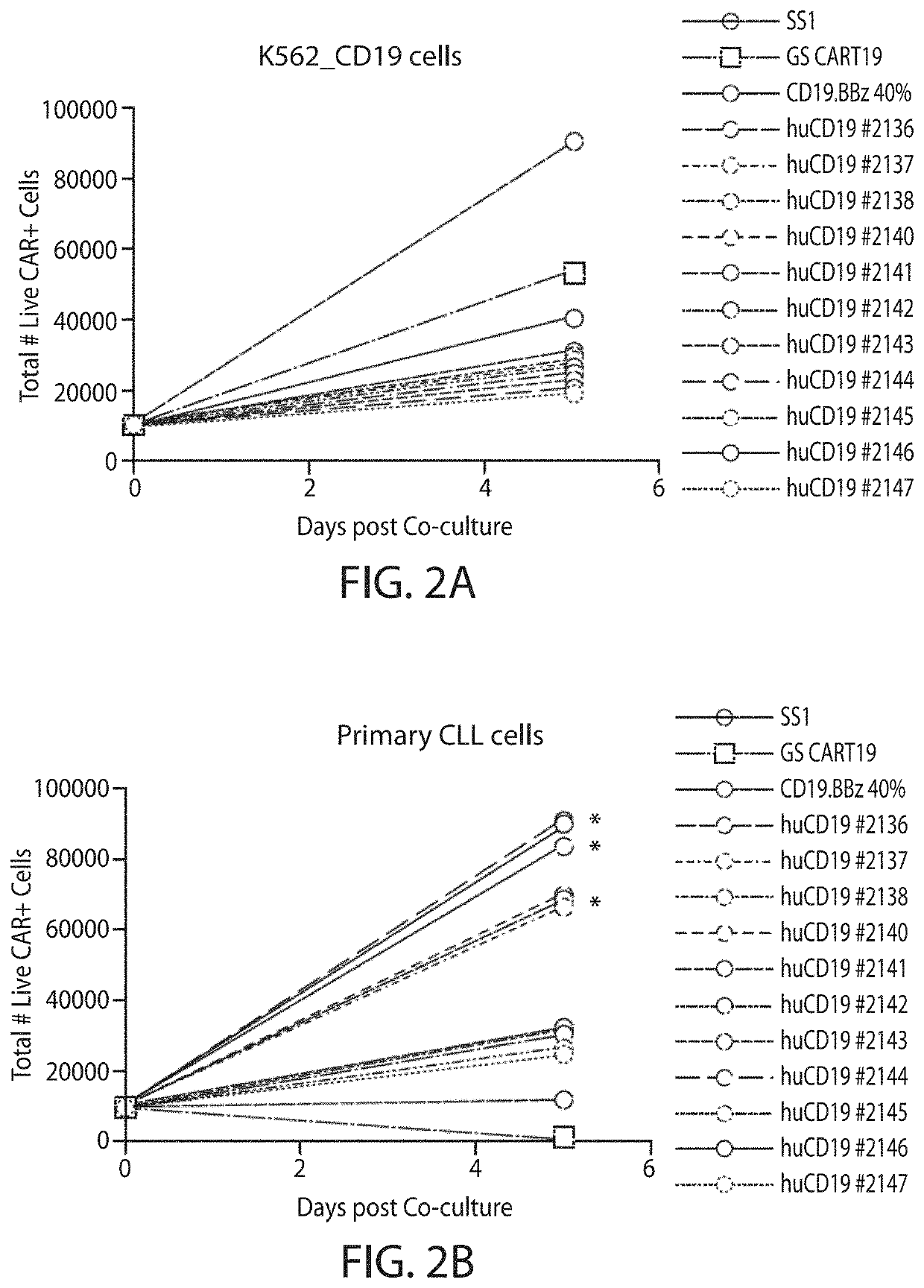
[0745]A CD19 antibody molecule can be, e.g., an antibody molecule (e.g., a humanized anti-CD19 antibody molecule) described in WO2014 / 153270, which is incorporated herein by reference in its entirety. Humanization of murine CD19 antibody is desired for the clinical setting, where the mouse-specific residues may induce a human-anti-mouse antigen (HAMA) response in patients who receive CART19 treatment, i.e., treatment with T cells transduced with the CAR19 construct. VH and VL sequences of hybridoma derived murine CD19 antibody were extracted from published literature (Nicholson et al, 1997, supra). Humanization was accomplished by grafting CDR regions from murine CD19 antibody onto human germline acceptor frameworks VH4_4-59 and VK3_L25 (vBASE database). In addition to the CDR regions, five framework residues, i.e. VH #71, #73, #78 and VL #71 #87, thought to support the structural integrity of the CDR regions were retained from the murine sequence. Fu...
example 2
ization of Anti-CD19 Soluble scFv Fragments Derived from Humanized CD19 IgG Antibodies
[0753]Soluble scFv fragments were generated from the humanized CD19 IgGs described in Example 1 using standard molecule biology techniques. These soluble scFvs were used in characterization studies to examine the stability, cell surface expression, and binding properties of the scFvs. Additionally, experiments were also conducted to investigate the impact of the potential PTM introduced during the humanization process.
scFv Expression and Purification
[0754]For transfection of each scFv construct, around 3e8 293F cells were transfected with 100 μg of plasmid using PEI as the transfection reagent at the ratio of 3:1 (PEI:DNA). The cells were grown in 100 ml EXPi293 Expression media (Invitrogen) in a shaker flask at 37° C., 125 rpm, 8% CO2. The culture was harvested after six days and used for protein purification.
[0755]293F cells were harvested by spinning down at 3500 g for 20 minutes. The supernatan...
example 3
Constructs
[0769]ScFv to be used in the final CAR construct were derived from the humanized IgG described in Example 1. The order in which the VL and VH domains appear in the scFv was varied (i.e., VL-VH, or VH-VL orientation), and where either three or four copies of the “G4S” (SEQ ID NO: 18) subunit, in which each subunit comprises the sequence GGGGS (SEQ ID NO:18) (e.g., (G4S)3 (SEQ ID NO:107) or (G4S)4 (SEQ ID NO:106)), connect the variable domains to create the entirety of the scFv domain, as shown in Table 2.
TABLE 2Humanized CD19 scFv constructs showing VH and VLorientation and linker length (“3G4S” is disclosed as SEQ ID NO: 107 and “4G4S” is disclosed as SEQ ID NO: 106).construct IDLength aaannotationVh changemscFvCTL019486VL-VH, 3G4S104879491VL-VH, 4G4SN / S104880491VL-VH, 4G4SN / Q104881491VH-VL, 4G4SN / S104882491VH-VL, 4G4SN / Q104875486VL-VH, 3G4SN / S104876486VL-VH, 3G4SN / Q104877486VH-VL, 3G4SN / S104878486VH-VL, 3G4SN / Q105974491VL-VH, 4G4SS / N105975491VH-VL, 4G4SS / N105976486VL-VH, ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| cycle length | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More