Indolyl and dihydroindolyl derivatives, their manufacture and use as pharmaceutical agents
a technology of dihydroindolyl and dihydroindolyl, which is applied in the field of nuclear hormone receptors, can solve the problems of limiting the therapeutic potential of this approach, unphysiologically high levels, and ultimately decreasing, and achieves the effects of reducing the requirement for exogenous insulin, restoring blood glucose and triglyceride levels, and moderate complian
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
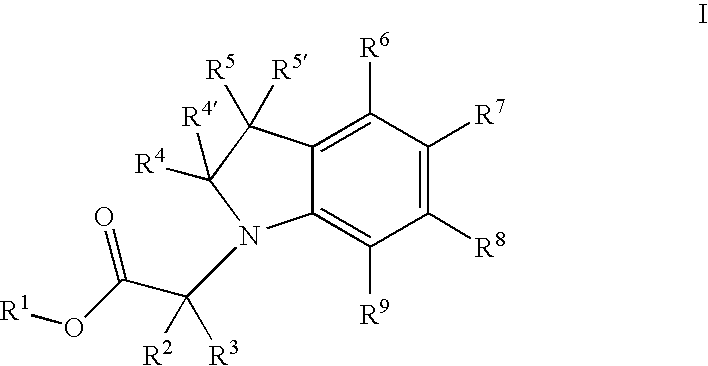
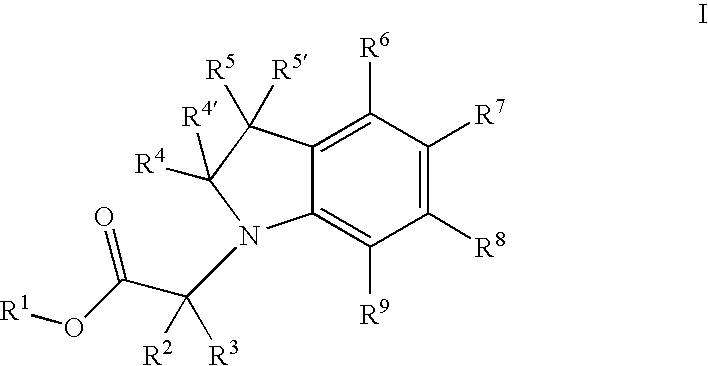
{5-[4-(2-Methoxy-ethyl)-2-(4-trifluoromethyl-phenyl)-pyrimidin-5-ylmethoxy]-indol-1-yl}-acetic acid
a] 5-(tert-Butyl-dimethyl-silanyloxy)-1H-indole
[0178]A solution of 5 g (37.55 mmol) of 5-hydroxy-indole, 6.13 g (39.4 mmol) of tert-butyldimethylsilyl chloride and 5.37 g (68.1 mmol) of imidazole in 50 ml DMF was stirred for 20 h at RT. The reaction mixture was taken up in ether, washed with 1N HCl and water and the ether solution was then concentrated under reduced pressure, to give 9.4 g (quant.) of clean 5-(tert-butyl-dimethyl-silanyloxy)-1H-indole.
[0179]MS: 248.1 (M+H)+.
b] [5-(tert-Butyl-dimethyl-silanyloxy)-indol-1-yl]-acetic acid ethyl ester
[0180]To a solution of 9.2 g (37.18 mmol) 5-(tert-butyl-dimethyl-silanyloxy)-1H-indole and 4.79 ml (40.9 mmol) ethyl bromoacetate in 140 ml of DMF was added 36.35 g (111.5 mmol) of cesium carbonate. The reaction mixture was stirred for 3 h at RT, taken up in ether and then washed with 1N HCl and water. The ether phase was concentrated under re...
example 2
{5-[4-Methyl-2-(4-trifluoromethyl-phenyl)-pyrimidin-5-ylmethoxy]-indol-1-yl}-acetic acid
a] 2-[1-Ethoxy-meth-(E,Z)-ylidene]-3-oxo-butyric acid ethyl ester
[0195]A solution of 5 g (38.42 mmol) 3-oxo-butyric acid ethyl ester, 12.78 ml (76.84 mmol) triethyl orthoformate in 80 ml acetic anhydride was boiled for 2.5 h at 150° C. The reaction mixture was concentrated under reduced pressure at 90° C. to provide 6.84 g of almost pure 2-[1-ethoxy-meth-(E,Z)-ylidene]-3-oxo-butyric acid ethyl ester.
[0196]MS: 186.9 (M+H)+.
b] 4-Methyl-2-(4-trifluoromethyl-phenyl)-pyrimidine-5-carboxylic acid ethyl ester
[0197]To a solution of 2 g (7.67 mmol) 4-trifluoromethyl-benzamidine HCl in 30 ml ethanol was added 0.76 g (7.67 mmol) sodium tert-butoxide and 1.43 g (7.67 mmol) 2-[1-ethoxy-meth-(E,Z)-ylidene]-3-oxo-butyric acid ethyl ester. The reaction was stirred at RT over night and for 1 h under reflux. After the solvent was removed under reduced pressure, the residue was taken up in ether and washed with 1N ...
example 3
{5-[4-Cyclopropyl-2-(4-trifluoromethyl-phenyl)-pyrimidin-5-ylmethoxy]-indol-1-yl}-acetic acid
a] (E,Z)-2-Cyclopropanecarbonyl-3-ethoxy-acrylic acid methyl ester
[0207]A solution of 10 g (70.34 mmol) 3-cyclopropyl-3-oxo-propionic acid methyl ester, 23.4 ml (140.68 mmol) of triethyl orthoformate in 100 ml acetic anhydride was refluxed at 150° C. for 5 h. The reaction mixture was concentrated at 95° C. under reduced pressure to give 14.35 g of crude (E,Z)-2-cyclopropanecarbonyl-3-ethoxy-acrylic acid methyl ester.
[0208]MS: 199.3 (M+H)+.
b] 4-Cyclopropyl-2-(4-trifluoromethyl-phenyl)-pyrimidine-5-carboxylic acid ethyl ester
[0209]To a solution of 4.74 g (18.19 mmol) 4-trifluoromethyl-benzamidine HCl in 50 ml of ethanol was added 1.82 g (18.19 mmol) of sodium tert-butoxide. After 2 min, 3.61 g of crude (E,Z)-2-cyclopropanecarbonyl-3-ethoxy-acrylic acid methyl ester was added and the reaction mixture was stirred over night at RT. The ethanol was removed under reduced pressure, the residue taken...
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperatures | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More