Quality control method of traditional Chinese medical preparation
A quality control method and technology of traditional Chinese medicine preparations, applied in the direction of medical formula, non-central analgesics, antipyretics, etc., can solve the problems of no quality control method for juvenile fengdan, so as to promote product sales and improve controllability The effect of improvement of sex and quality standards
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0059] Below in conjunction with the examples, the present invention is further described, the following examples are illustrative, not limiting, and the protection scope of the present invention cannot be limited by the following examples.
[0060] A kind of quality control method of traditional Chinese medicine preparation Baoyouhuafengdan, the steps of its method are:
[0061] (1) Microscopic identification:
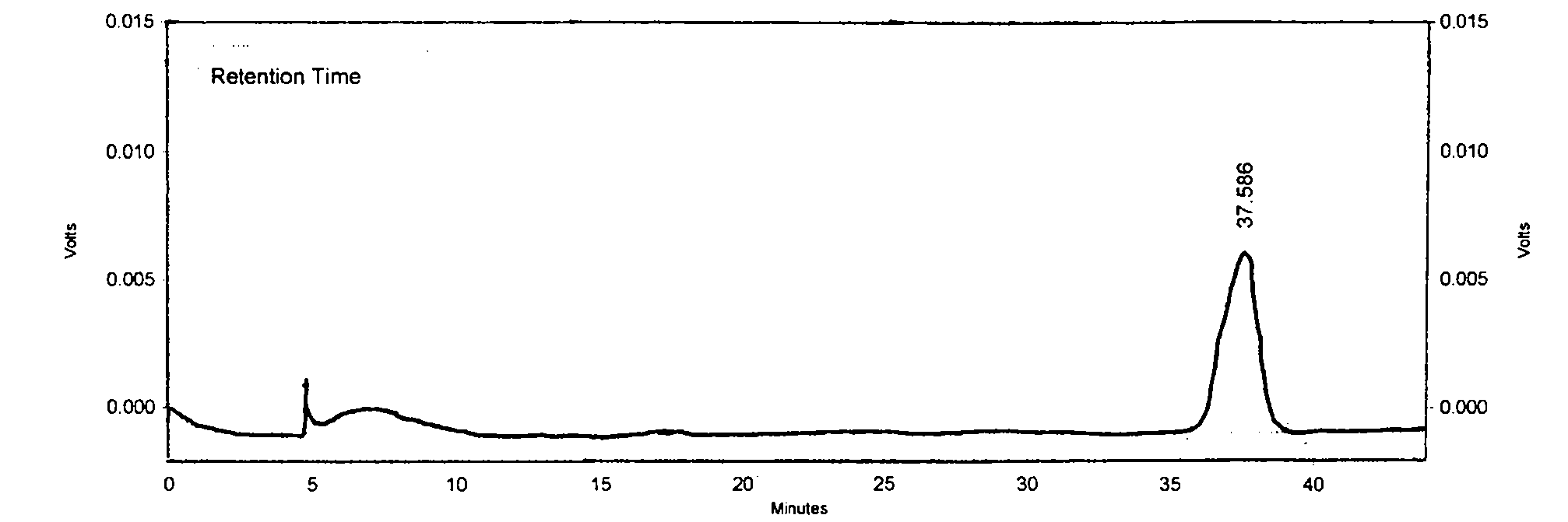
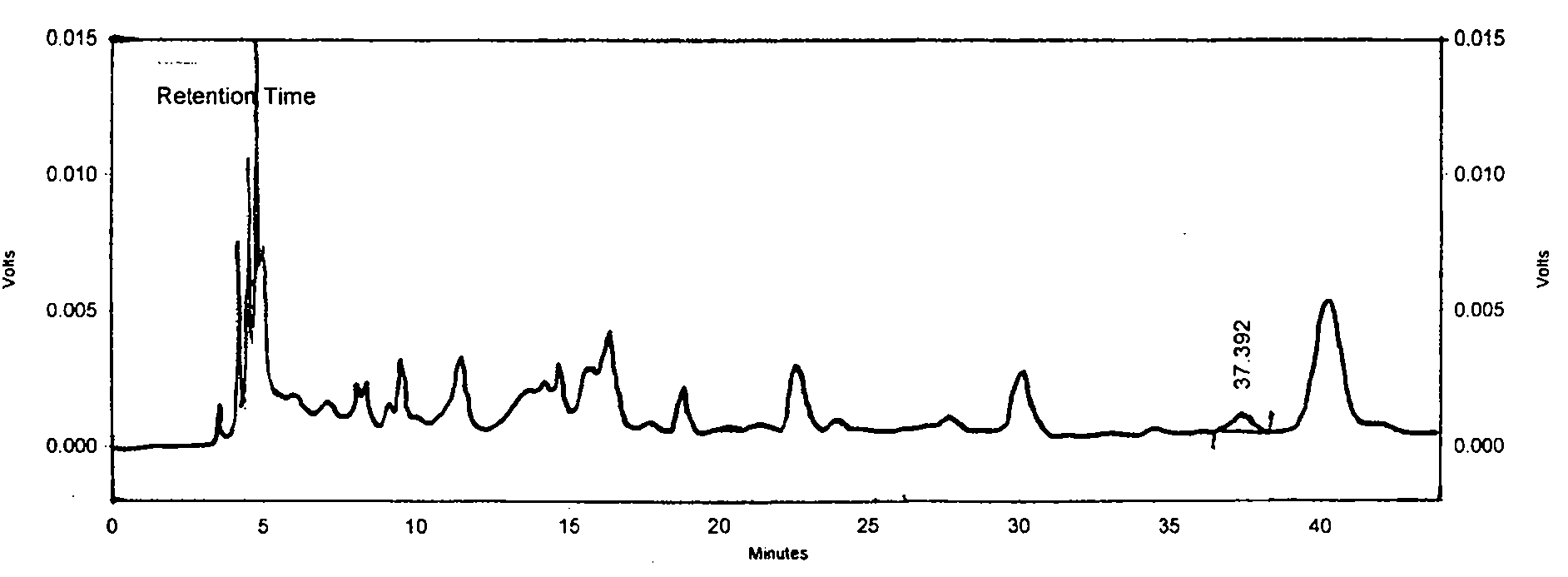
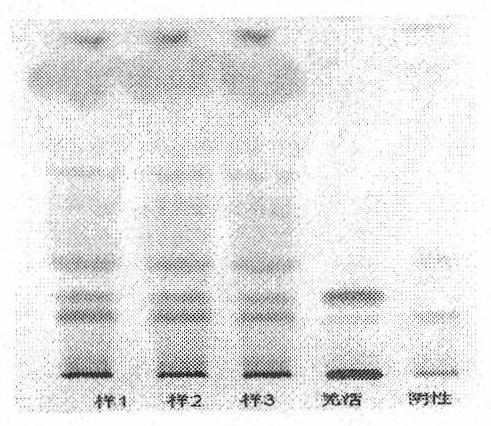
[0062] The microscopic description is as follows: Since this product is raw powder used as medicine, the diameter of calcium oxalate cluster crystals is 20-68 μm, with sharp edges and corners; the parenchyma cells around the fiber bundles contain square crystals of calcium oxalate, forming crystal fibers; the thick-walled cells are polygonal or long polygonal , 70-180 μm in diameter, thick wall, slightly lignified, and obvious pits; hesperidin crystals are slightly yellowish, present in mesophyll tissue, especially in palisade cells; tubules contain brown-yellow secre...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Diameter | aaaaa | aaaaa |
| Diameter | aaaaa | aaaaa |
| Diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com