Antitumor drug with double active components and application thereof
A technology of anti-tumor drugs and active ingredients, applied in the field of anti-tumor drugs, can solve the problems of unsatisfactory tumor treatment effects, and achieve clear and significant anti-tumor effects, significant anti-tumor activity, and enhanced inhibitory effects
Active Publication Date: 2011-09-28
MEDICINE & BIOENG INST OF CHINESE ACAD OF MEDICAL SCI +1
View PDF3 Cites 5 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0023] Experiments have proved that the effect of using the above drugs alone is not very satisfactory for tumor treatment. Therefore, how to choose appropriate drugs and use them in combination to improve the effect of treating tumors is an important research topic in this field.
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0061] Recipe: Dipyridamole is 1mg, 5mg, 50mg, 100mg, 200mg, 400mg, 800mg or 1600mg respectively, Ubenimex is 1mg, 10mg, 50mg, 100mg, 200mg, 400mg, 800mg, 1600mg or 3200mg respectively. Pyridamole and Ubenimex are stirred and mixed.
Embodiment 2
[0063] Recipe: dipyridamole is 1mg, 5mg, 50mg, 100mg, 200mg, 400mg, 800mg or 1600mg respectively, and dexamethasone is 0.1mg, 1mg, 10mg, 20mg, 40mg, 80mg or 160mg respectively. Stir dipyridamole and dexamethasone together.
Embodiment 3
[0065] Recipe: Ubenimex is 1mg, 10mg, 50mg, 100mg, 200mg, 400mg, 800mg, 1600mg or 3200mg respectively, and dexamethasone is 0.1mg, 1mg, 10mg, 20mg, 40mg, 80mg or 160mg respectively. Stir Ubenimex and Dexamethasone to mix.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
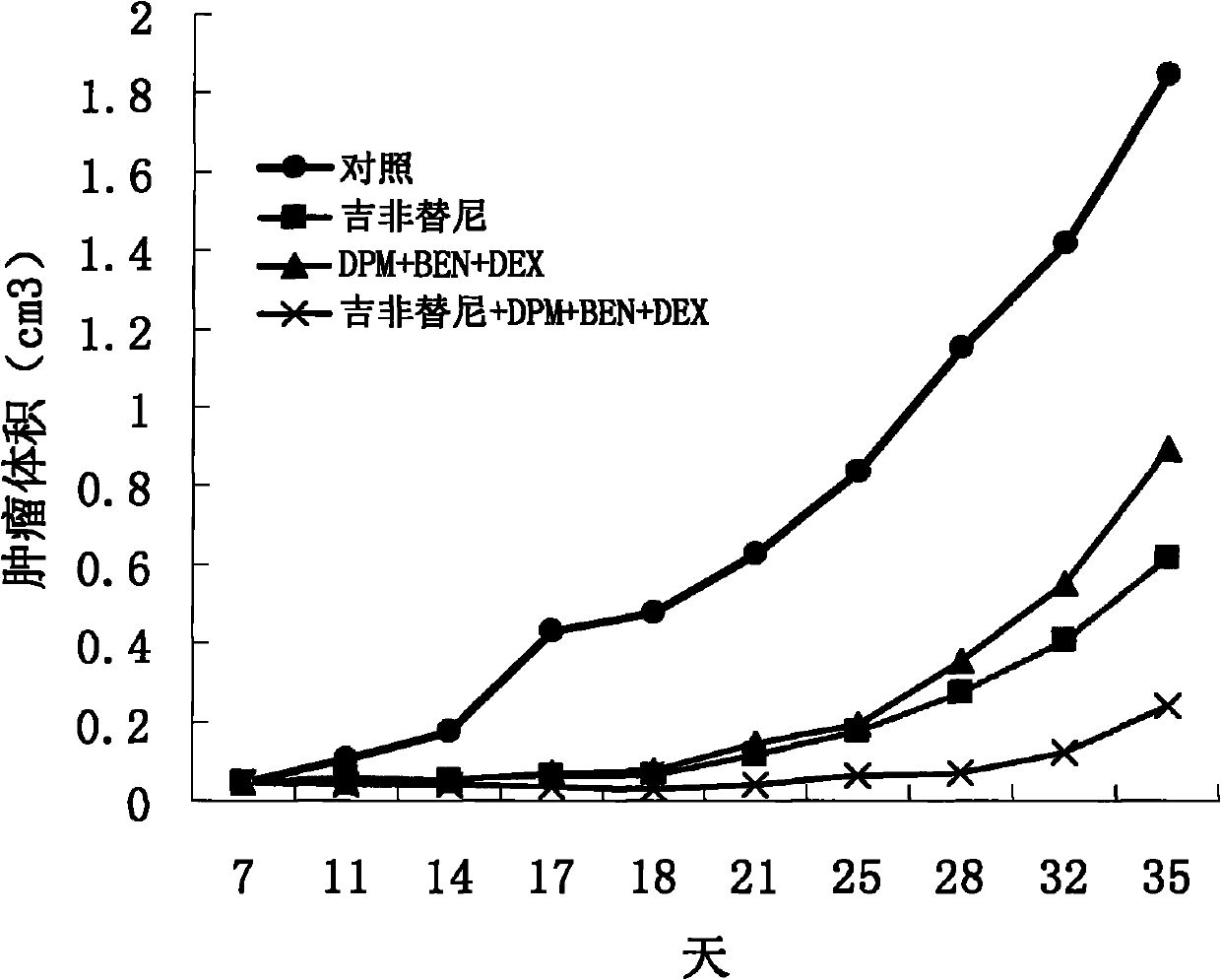
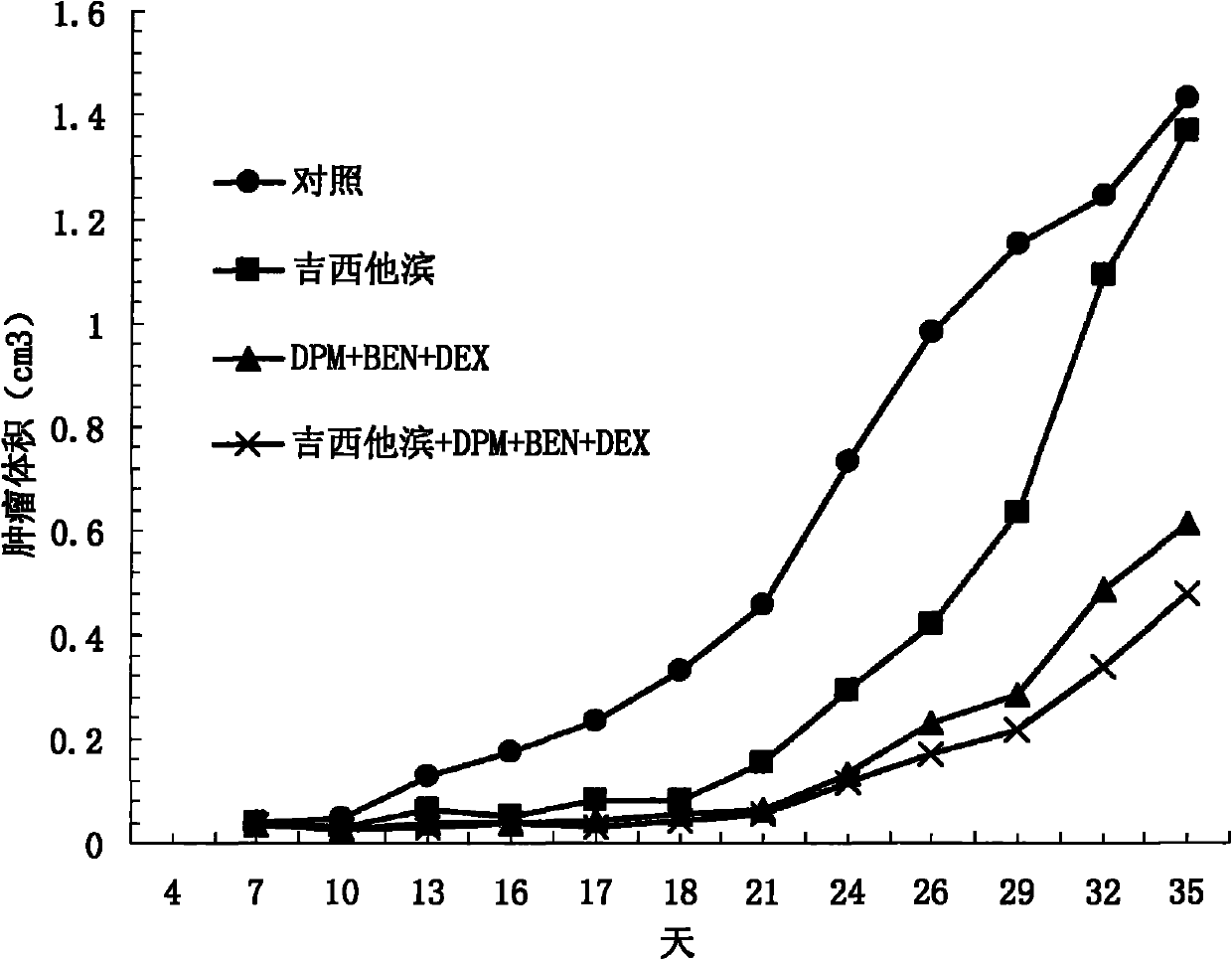
The invention discloses an antitumor drug with double active components, and an application thereof. The antitumor drug comprises an independent first active component and an independent second active component. The independent first active component comprises at least two of A, B and C. The A comprises Dipyridamole and pharmaceutically acceptable derivatives of Dipyridamole or pharmaceutically acceptable analogues of Dipyridamole or pharmaceutically acceptable salts of Dipyridamole. The B comprises Ubenimex bestatin and pharmaceutically acceptable derivatives of Ubenimex bestatin or pharmaceutically acceptable analogues of Ubenimex bestatin or pharmaceutically acceptable salts of Ubenimex bestatin. The C comprises Dexamethasone and pharmaceutically acceptable derivatives of Dexamethasone or pharmaceutically acceptable analogues of Dexamethasone. The independent second active component comprises Taxol, ADM, DDP, MMC, 5-FM, Gemcitabine or a tyrosine kinase inhibitor. The antitumor drug provided by the invention can effect on multiple targets and multiple paths, and thus antineoplastic effects of the antitumor drug are clear and obvious.
Description
technical field [0001] The invention relates to a medicine with antitumor effect, in particular to an antitumor medicine comprising dipyridamole, ubenimex and dexamethasone and an antitumor chemotherapeutic medicine. Background technique [0002] The toxicity of currently clinically used antitumor drugs is a prominent problem in tumor chemotherapy. Finding its own low toxicity and acting on specific links or molecular targets in the tumor microenvironment to improve the anti-tumor effect or reduce its toxicity has become a new way to study anti-tumor drugs, and it is also an urgent need for clinical treatment of tumors. [0003] Malignant tumor is a common disease that seriously threatens human health. In today's world, there is an urgent need for anti-tumor drugs. According to the data in 2007, there were 12 million newly diagnosed cancer patients in the world, and 7.6 million people died of cancer. According to the data of the top ten disease mortality and cause of deat...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K33/24A61K31/704A61K31/573A61K31/519A61K31/513A61K31/407A61K31/337A61K31/198A61K45/00A61P35/00
Inventor 刘秀均甄永苏郑艳波李毅吴淑英
Owner MEDICINE & BIOENG INST OF CHINESE ACAD OF MEDICAL SCI