Medicine for treating hyperplasia of mammary glands, mastodynia and general mammary diseases and preparation method thereof
A technology for mammary gland hyperplasia and mammary gland diseases, which can be applied in the direction of diseases, drug combinations, and pharmaceutical formulas, and can solve the problems of high price and limited wide application
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
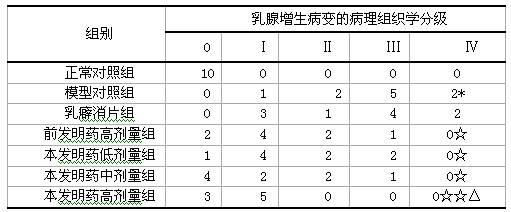
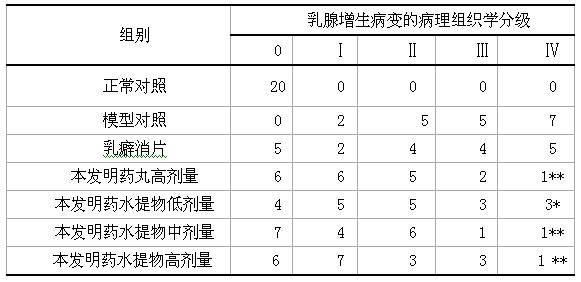
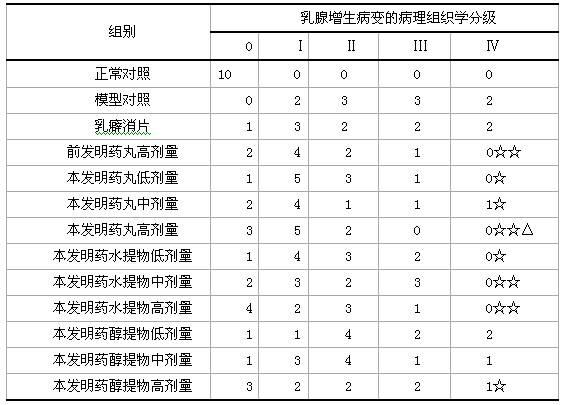
Examples
Embodiment 1
[0138] 15 grams of Angelica, 9 grams of Yuanhu, 15 grams of Fritillaria, 12 grams of Wangbuliuxing, 12 grams of Rhizoma Cyperi, 12 grams of Curcuma, 9 grams of Bupleurum, 10 grams of Turtle Shell, 12 grams of Atractylodes macrocephala, 9 grams of Woody , Poria cocos 9 grams, saponins 6 grams, motherwort 15 grams.
Embodiment 2
[0140] 15 grams of Angelica, 12 grams of Yuanhu, 9 grams of Fritillaria, 9 grams of Wangbuliuxing, 15 grams of Cyperus Cyperi, 9 grams of Curcuma, 15 grams of Bupleurum, 15 grams of Turtle Shell, 9 grams of Atractylodes macrocephala, 12 grams of Woody Fragrance , Poria cocos 12 grams, saponins 9 grams, motherwort 9 grams, cistanche 15 grams.
Embodiment 3
[0142] 10 grams of Angelica, 12 grams of Yuanhu, 15 grams of Fritillaria, 15 grams of Wangbuliuxing, 9 grams of Rhizoma Cyperi, 15 grams of Curcuma, 9 grams of Bupleurum, 10 grams of Turtle Shell, 15 grams of Atractylodes macrocephala, and 15 grams of woody fragrance , 15 grams of Poria cocos, 6 grams of saponins, 12 grams of motherwort, 9 grams of cistanche, 9 grams of antler cream, and 9 grams of epimedium.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More