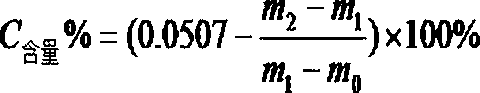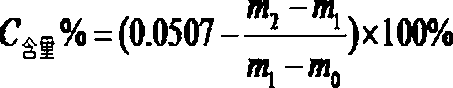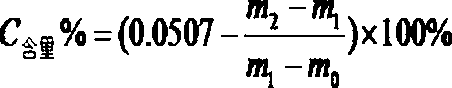Method for measuring carbon content in lithium iron phosphate
A lithium iron phosphate and determination method technology, applied in the direction of weighing by removing certain components and preparing test samples, can solve the problems of high detection cost and easy errors, and achieve low consumption of consumables and high accuracy , the effect of simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] (1) Put the cleaned porcelain boat into a high-temperature box-shaped electric furnace, burn it at 800°C for 2 hours, then cool it down to 200°C, transfer the porcelain boat to a desiccator, wait to cool to room temperature, weigh and record m0 =17.6269g.
[0042] (2) Spread 1g of the material in the fired porcelain boat, put it in a vacuum drying oven, and dry it at 120°C for 2 hours. After cooling down to room temperature, weigh the porcelain boat and record the total weight of the dried material is m1= 18.5616g;
[0043] (3) Put the porcelain boat and materials dried in step 2 into a high-temperature box-shaped electric furnace, burn at 800°C for 2 hours, then cool down to 200°C, transfer the porcelain boat to a desiccator, and wait to cool to room temperature. Weigh and record m2= 18.5795g;
[0044] (4) According to the weighing result, calculate the mass fraction of carbon element according to the announcement, where m0 is the weight of the empty porcelain boat, ...
Embodiment 2
[0049] (1) Put the cleaned porcelain boat into a high-temperature box-shaped electric furnace, burn it at 1000°C for 2 hours, then cool it down to 200°C, transfer the porcelain boat to a desiccator, wait to cool to room temperature, weigh and record m0 =18.3056g;
[0050] (2) Spread 1.2g of the material in the fired porcelain boat, put it in a vacuum drying oven, and dry it at 150°C for 2 hours. After cooling down to room temperature, weigh the total weight of the porcelain boat and the dried material Recorded as m1=19.472g;
[0051] (3) Put the porcelain boat and materials dried in step 2 into a high-temperature box-shaped electric furnace, burn at 1000°C for 2 hours, then cool down to 200°C, transfer the porcelain boat to a desiccator, and wait to cool to room temperature. Weigh and record m2=19.505g;
[0052] (4) According to the weighing result, calculate the mass fraction of carbon element according to the announcement, where m0 is the weight of the empty porcelain boat...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com