N(2)-L-alanyl-L-glutamine/compound amino acid injection (18AA-V) pharmaceutical composite preparation
A 18AA-V, compound amino acid technology, applied in the directions of drug combination, drug delivery, pharmaceutical formulation, etc., can solve the problems of not exceeding 3.5%, failing to meet clinical requirements, etc., achieving less adverse reactions, good application prospects, and scope of application wide effect
Active Publication Date: 2013-05-01
ZHEJIANG CHANGDIAN PHARMA
View PDF2 Cites 4 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0003] Since N(2)-L-alanyl-L-glutamine has the function of promoting positive nitrogen balance, and its solution is a hypertonic solution, the maximum concentration of the infusion solution should not exceed 3.5%. , there will be serious adverse reactions such as nitrogen loss, vascular stimulation and dehydration of vital organs, which will cause N(2)-L-alanyl-L-glutamine to fail to meet clinical requirements
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
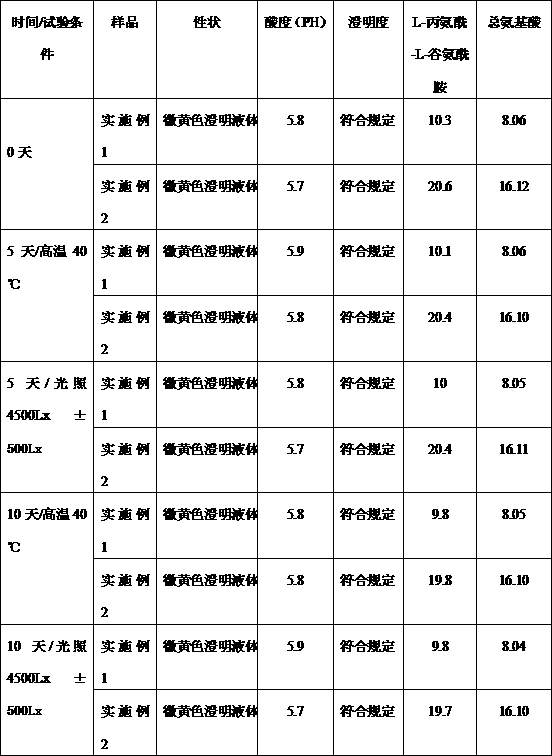
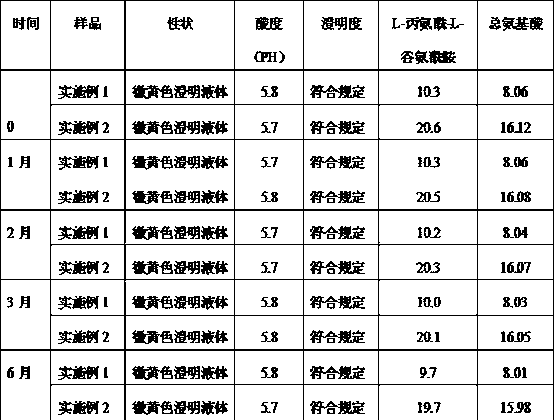
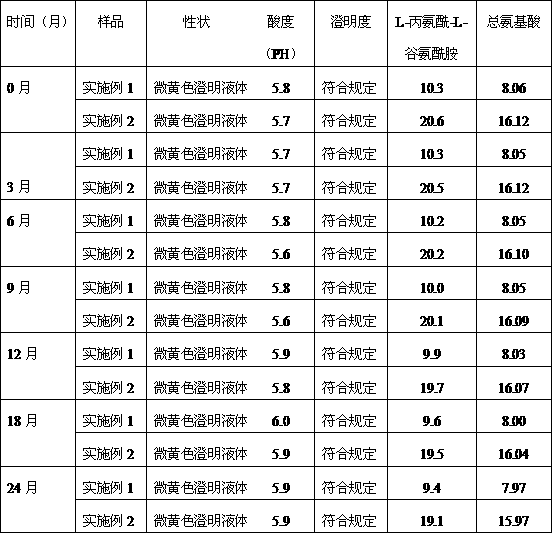
[0012] N(2)-L-alanyl-L-glutamine / compound amino acid injection (18AA-V) drug combination preparation is made of the following components by weight:
[0013] N(2)-L-alanyl-L-glutamine for injection 10g;
[0014] Sterile water for injection 50ml;
[0015] Compound amino acid injection (18AA-V) 250ml: 8.06g (total amino acid) and 12.5g xylitol.
Embodiment 2
[0017] N(2)-L-alanyl-L-glutamine / compound amino acid injection (18AA-V) drug combination preparation is made of the following components by weight:
[0018] N(2)-L-alanyl-L-glutamine for injection 20g;
[0019] Sterile water for injection 100ml;
[0020] Compound amino acid injection (18AA-V) 500ml: 16.12g (total amino acid) and 25g xylitol.
[0021] Quality Research Survey
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to the technical field of medicines and discloses an N(2)-L-alanyl-L-glutamine / compound amino acid injection (18AA-V) pharmaceutical composite preparation which is prepared by mixing N(2)-L-alanyl-L-glutamine and compound amino acid injection (18AA-V). The pharmaceutical composite preparation has the advantages of definite components, stable quality, high bioavailability, less adverse reactions and the like and has a good application prospect in clinical practice.
Description
technical field [0001] The invention relates to the technical field of medicine, in particular to an N(2)-L-alanyl-L-glutamine / compound amino acid injection (18AA-V) drug combination preparation. Background technique [0002] N(2)-L-alanyl-L-glutamine is a component of parenteral nutrition, which can promote positive nitrogen balance, regulate muscle protein synthesis, and prevent intestinal mucosal passage caused by long-term parenteral nutrition. Increased permeability and atrophy, C can enhance immune function and prevent the occurrence of bacteremia. N(2)-L-alanyl-L-glutamine can be decomposed into alanine and glutamine in the body, and glutamine can be supplemented by parenteral nutrition infusion. [0003] Since N(2)-L-alanyl-L-glutamine has the function of promoting positive nitrogen balance, and its solution is a hypertonic solution, the maximum concentration of the infusion solution should not exceed 3.5%. , there will be serious adverse reactions such as nitrogen...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K38/05A61K9/08A61P3/02A61K31/4172A61K31/405A61K31/401A61K31/198
Inventor 傅苗青历达中陈宇东
Owner ZHEJIANG CHANGDIAN PHARMA