Method for on-line synthesizing saccharose-6-laurate by lipase catalysis
A technology of laurate and lipase, applied in biochemical equipment and methods, biochemical instruments, biochemical cleaning devices, etc., can solve the problems of long reaction time, difficult separation, low conversion rate and selectivity, and achieve high reaction Effects of selectivity, shortened reaction time, and high conversion
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0027] Embodiment 1: the synthesis of sucrose-6-laurate
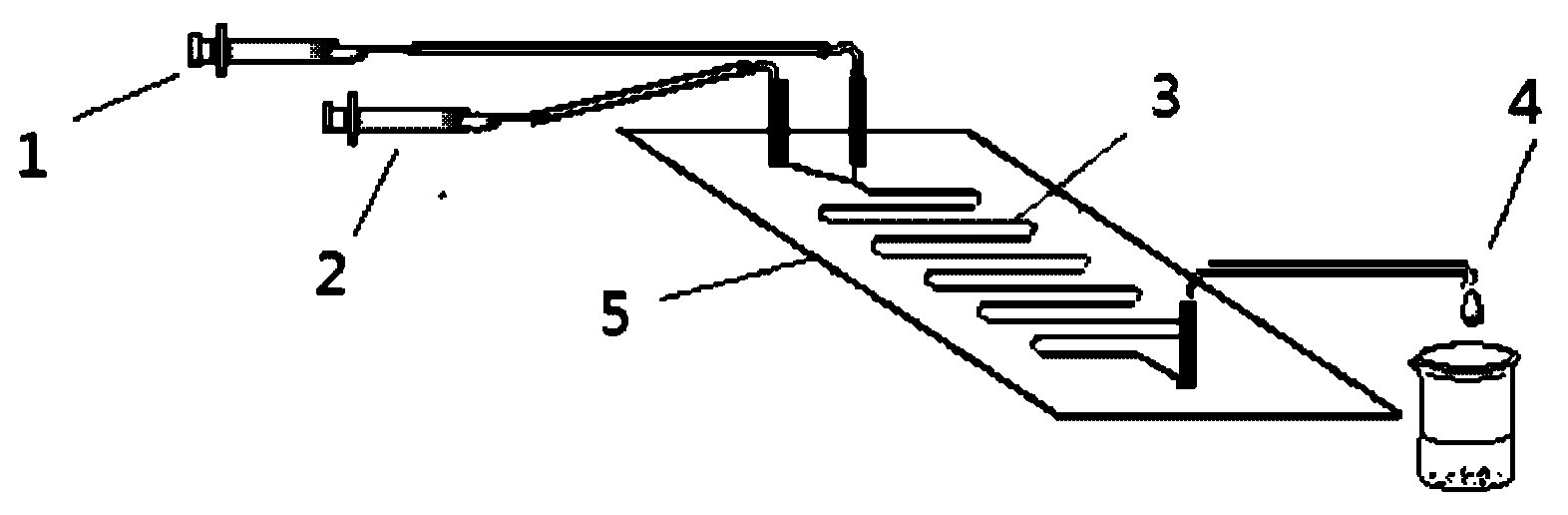
[0028] refer to figure 1 , sucrose (0.4mmol) was dissolved in 10mL (tert-amyl alcohol: DMSO = 4:1 (v / v)) mixed solvent, vinyl laurate (4.4mmol) was dissolved in 10mL tert-amyl alcohol, and then packed separately Prepare in a 10mL syringe. 0.87g of lipase Lipozyme TLIM was evenly filled in the reaction channel, and driven by the PHD2000 syringe pump, the two reaction solutions were mixed with 10.4μLmin -1The flow rate enters the reaction channel through the "Y" joint to carry out the acylation reaction. The temperature of the reactor is controlled at 52°C through a water bath thermostat, and the reaction solution flows continuously in the reaction channel for 30 minutes. The reaction solution is collected online by the product collector, and the pressure is reduced The solvent was distilled off, and the column was wet-packed with 200-300 mesh silica gel. The eluent was chloroform:methanol=10:1, the column height was 35...
Embodiment 2-5
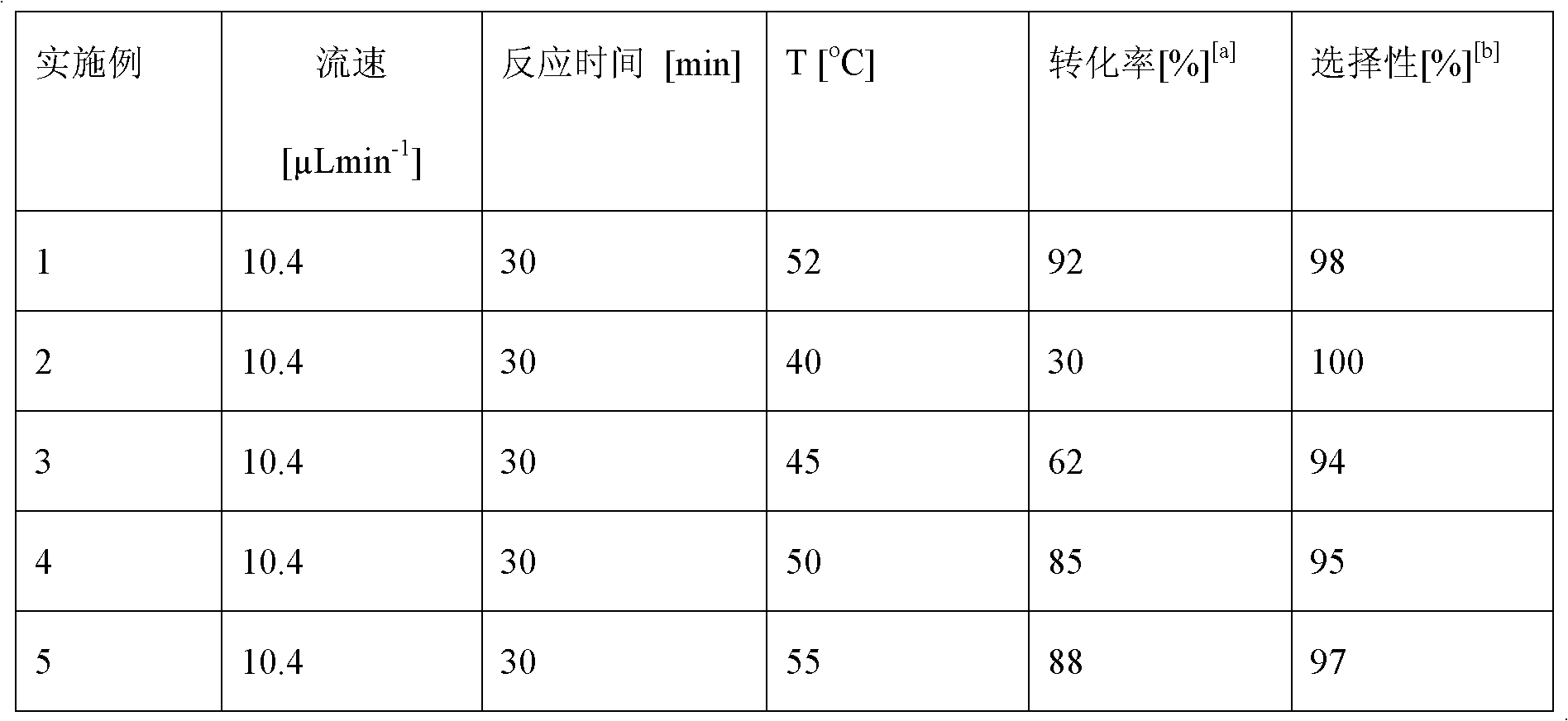
[0032] Change the temperature of the microfluidic channel reactor, others are the same as in Example 1, and the reaction results are as shown in Table 1:
[0033] Table 1: Effect of Temperature on Reaction
[0034]
[0035] [a]: The conversion rate was determined by HPLC; [b]: The content of sucrose-6-laurate in the total sucrose ester product
[0036] The results in Table 1 show that when the flow rate is 10.4μLmin -1 , when the reaction time is 30min, the conversion rate increases obviously with the increase of the reaction temperature. When the reaction temperature reaches 52°C, the conversion rate and selectivity of the reaction are the best. The decrease in enzyme activity leads to a decrease in the conversion rate and selectivity of the reaction, so the optimal reaction temperature of sucrose-6-laurate in the microfluidic channel reactor is 52°C.
Embodiment 6-9
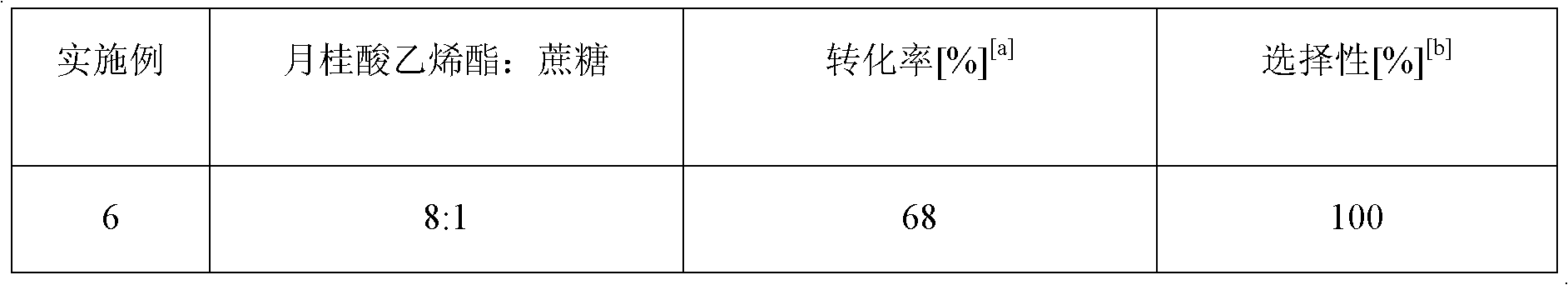
[0038] Change the substrate molar ratio of vinyl laurate and sucrose in the microfluidic microchannel reactor to be 8: 1 (Example 6), 9: 1 (Example 7), 10: 1 (Example 8), 12: 1 (embodiment 9), other are with embodiment 1, and the result is as shown in table 2.
[0039] Table 2: Effect of sucrose to vinyl laurate substrate ratio on the reaction
[0040]
[0041]
[0042] [a]: conversion rate is measured by HPLC; [b]: the result of the content table 2 of sucrose-6-laurate in the total sucrose ester product shows that, along with the increase of reactant vinyl laurate, the conversion rate of reaction also increases With the increase, when the substrate ratio is 11:1, the conversion and selectivity of the reaction are optimal. Now if continue to increase the consumption of reactant vinyl laurate, will cause the selectivity of reaction to reduce, thereby, the optimal substrate ratio of this reaction is 11:1.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More