Pharmaceutical composition of salvia miltiorrhiza extractive, and use thereof
A technology of composition and extract, applied in the pharmaceutical composition of Danshen extract and its application field
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
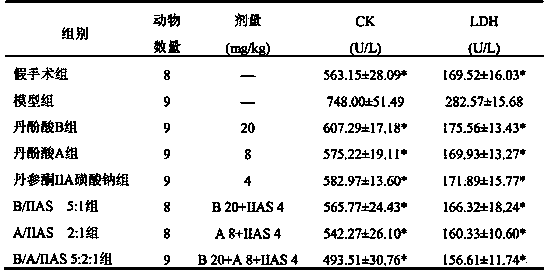
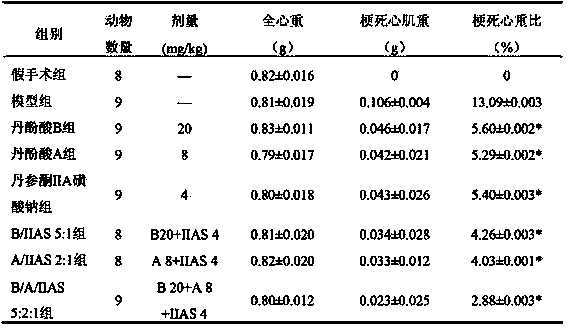
[0112] Example 1: Lyophilized preparation of salvianolic acid B, salvianolic acid A and sodium tanshinone IIA sulfonate (5:2:1)
[0113] Get 20g of salvianolic acid B, 8g of salvianolic acid A and 4g of sodium tanshinone IIA sulfonate in Example 1, add 32g of sodium ascorbate and 12.5g of mannitol, dissolve in 350ml of distilled water, and set the volume to 400ml, filter and sterilize, aseptic conditions The next part is packed in 100 vials, freeze-dried, and sealed under nitrogen to obtain the final product.
Embodiment 2
[0114] Example 2: Lyophilized preparation of salvianolic acid B, salvianolic acid A and sodium tanshinone IIA sulfonate (3:1:1)
[0115] Get 21g of salvianolic acid B, 7g of salvianolic acid A and 7g of sodium tanshinone IIA sulfonate in Example 1, add 7.4g of magnesium hydrogen phosphate, 35g of sodium ascorbate, and 12.5g of mannitol, dissolve in 250ml of distilled water, stir for 20 minutes, and constant volume to 300ml, filter out the insoluble matter, sterilize by filtration, subpackage in 100 vials under aseptic conditions, freeze-dry, and seal under nitrogen to obtain the product.
Embodiment 3
[0116] Example 3: Salvianolic acid B, salvianolic acid A and sodium tanshinone IIA sulfonate (9:3:2) freeze-dried powder injection
[0117] Take 18g of salvianolic acid B, 6g of salvianolic acid and 4g of sodium tanshinone IIA sulfonate, add 6g of magnesium hydrogen phosphate, 27g of sodium ascorbate, and 12.5g of mannitol, dissolve them in 250ml of distilled water, stir for 20 minutes, filter out insolubles, filter out sterilized, freeze-dried under aseptic conditions, the newly prepared powdery pharmaceutical composition is divided into 100 vials, sealed under nitrogen, and the product is obtained.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com