Preparation method of 1,2-benzisothiazole-3-ketone
A technology of benzisothiazole and sodium hypochlorite, applied in the direction of organic chemistry, to achieve the effects of short time, high yield and low cost
Inactive Publication Date: 2016-07-27
SHANXI UNIV
View PDF4 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
The classic preparation method of 1,2-Benzosoxazoline-3-ones is to use either thionyl chloride at low temperature or 1,1'-carbonyldiimidazole (CDI) at high temperature, which has its limitations
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
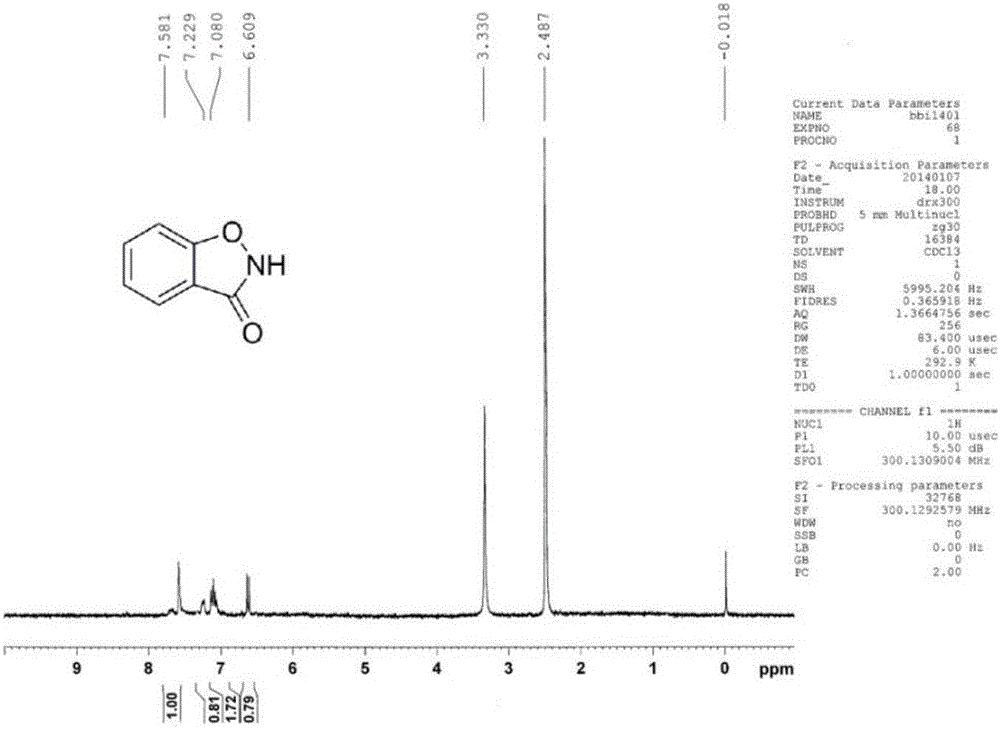
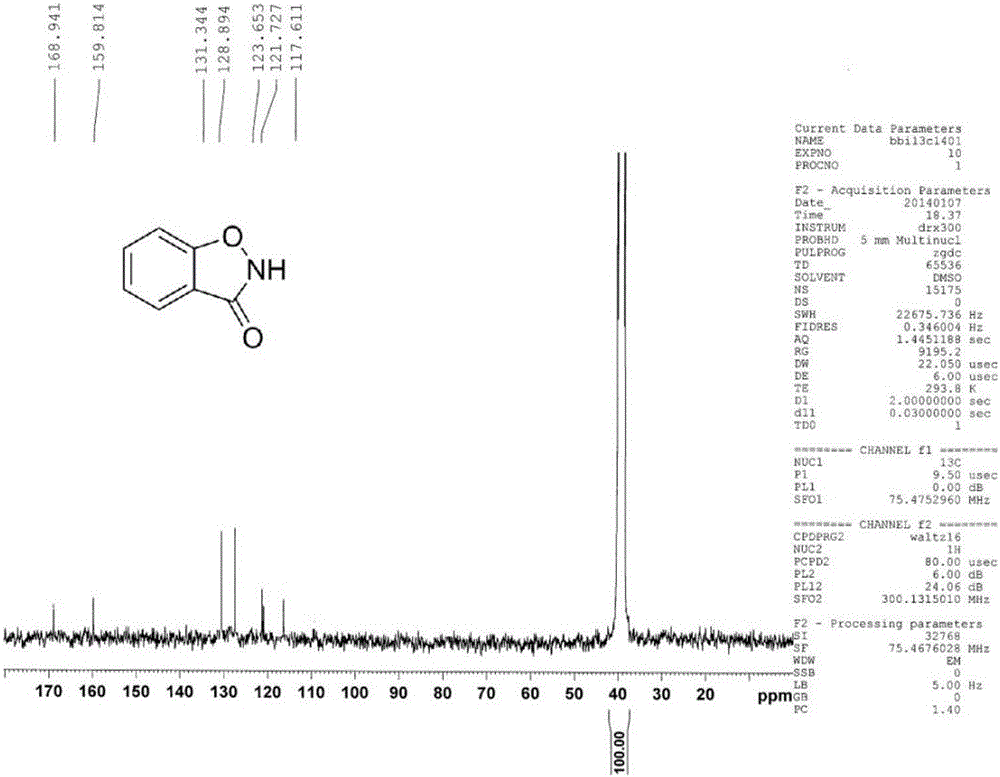
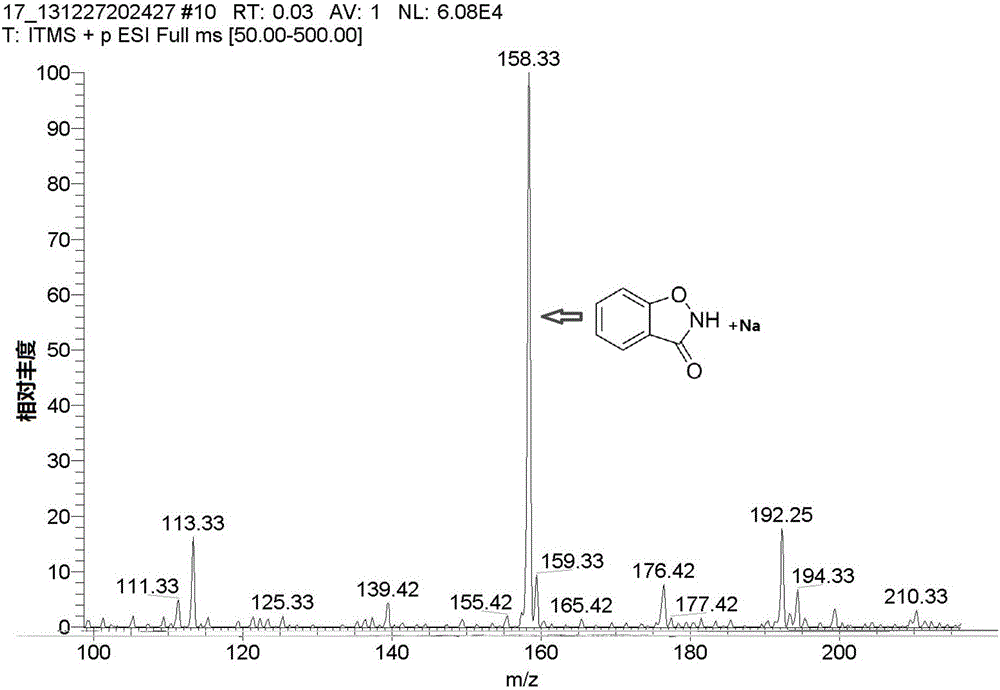
[0015] Dissolve salicyl hydroxamic acid (153mg, 1mmol) in 2ml of methanol, add 6.7mL (10mmol) of 10% sodium hypochlorite, react at room temperature for 30s, dilute with distilled water, and extract three times with ethyl acetate, the organic phase Evaporate to dryness to obtain a light yellow solid, and obtain 110 mg of the product 1,2-benzisothiazol-3-one after suction filtration (yield 82%).
[0016] Product characterization see attached figure 1 , 2 , 3.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention provides a preparation method of 1,2-benzisothiazole-3-ketone. The preparation method comprises the following steps of dissolving salicylhydroxamic acid into methanol; adding sodium hypochlorite with the concentration being 10 to 20 percent; taking a reaction for 20 to 40s at room temperature; performing dilution by distilled water; performing extraction by ethyl acetate; performing organic phase drying by distillation to obtain slight yellow solid; performing suction filtration to obtain a product of the 1,2-benzisothiazole-3-ketone. The method has the advantages that the synthesis process is simple; the cost is low; green and environment-friendly effects are achieved; the time is short; no byproduct is produced; the yield is as high as more than 82 percent.
Description
technical field [0001] The invention relates to a preparation method of heterocyclic compounds, in particular to a preparation method of 1,2-benzisothiazol-3-ones (1,2-Benzisoxazoline-3-ones). Background technique [0002] Green and sustainable synthesis is the development trend of modern organic chemistry, and the main goal of current organic synthesis is to develop "green synthetic methods" and quickly obtain target compounds. In fact, the preparation of heterocyclic compounds is a persistent problem in organic synthesis. Heterocyclic compounds occupy an important position in coordination chemistry, biochemistry, pharmacology, material science, etc. Many biologically active natural products are heterocyclic compounds and are used in pesticides, pharmaceuticals and fragrances. For example, 1,2-benzisoxazole substituted compounds are used for anticancer, antimicrobial, antithrombotic. The classic preparation method of 1,2-Benzisoxazoline-3-ones is to use either thionyl chl...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07D261/20C07D275/04
CPCC07D261/20C07D275/04
Inventor 霍方俊阴彩霞
Owner SHANXI UNIV