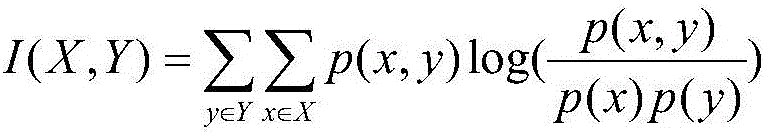Method for screening tumor protein markers on basis of multilayer complex network
A complex network and protein technology, applied in the field of screening tumor protein markers based on multi-layer complex networks, can solve problems such as failure to learn, difficult to interpret output results, and long learning time for artificial neural network algorithms, and the method is simple and accurate. high degree of effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
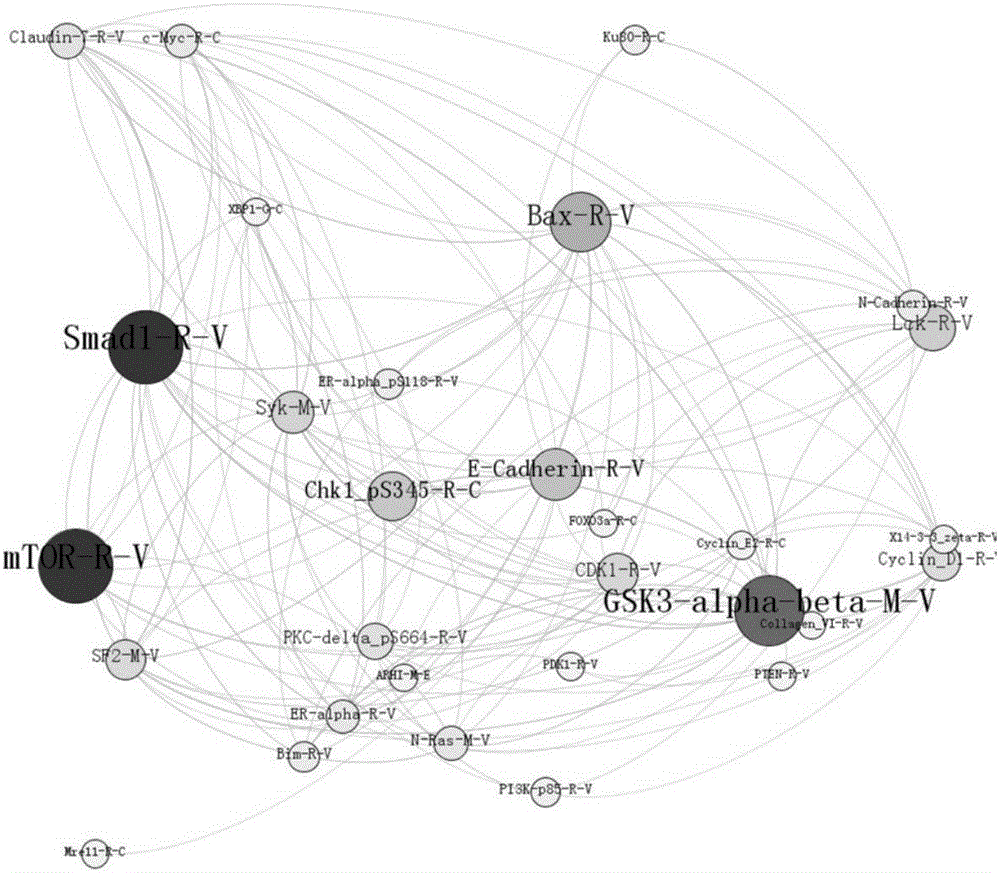
[0067] The source of the research data is The Cancer Genome Atlas / TCGA (https: / / tcga-data.nci.nih.gov / tcga / dataAccessMatrix.htm). Select the data of invasive breast cancer patients whose protein Experssion-Protein data level is 3 to download. Among them, there are 285 protein data from 937 patients. Among the protein expression data, 45 are normal tissue protein data of breast cancer patients, and the rest are tumor tissue protein data of breast cancer patients. In the protein data of normal tissues and tumor tissues, there are many proteins that are not expressed or have a low expression rate. After removing individuals with no protein expression, the protein data of normal tissues and breast tumor tissues with a size of 137×45 are obtained, that is, 137 out of 45 patients There are three different types of normal tissue protein data and 137 same types of tumor tissue protein data in normal tissues.
[0068] The random forest model is used to encapsulate and filter the protein...
Embodiment 2
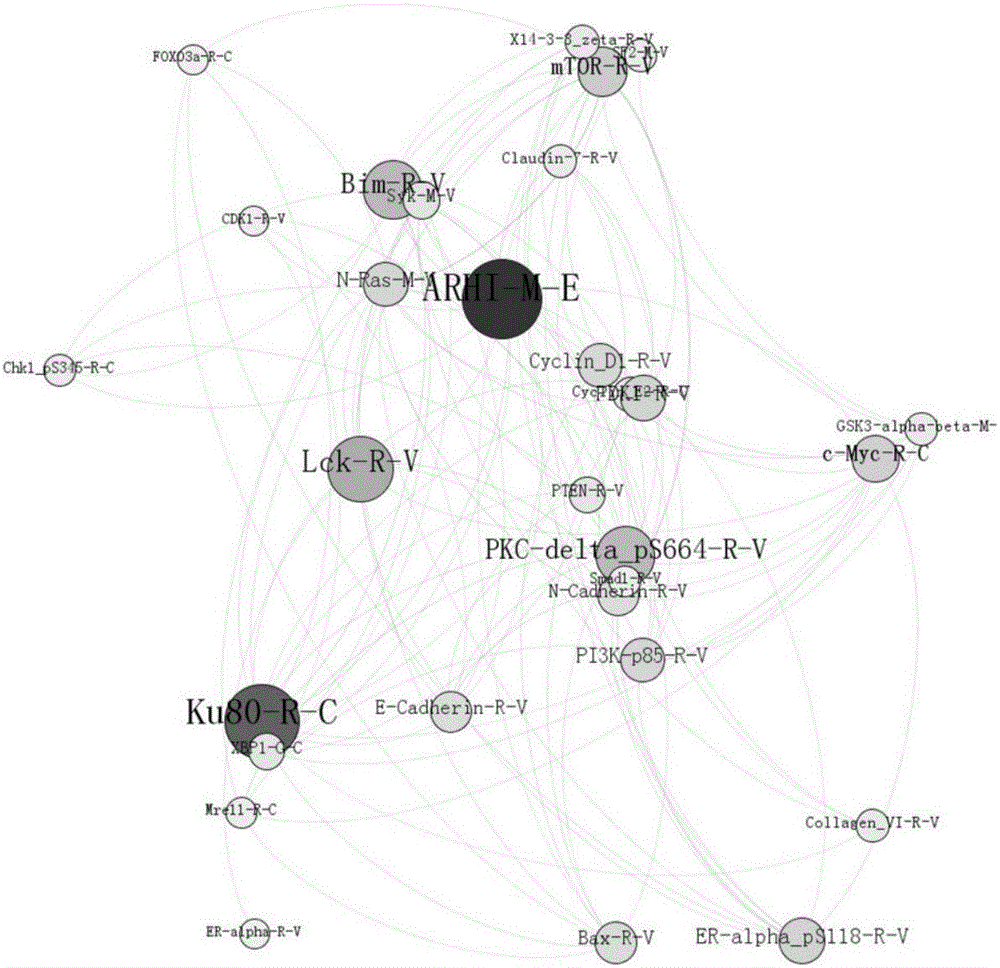
[0077] The method of Example 1 was used to download the protein data of lung cancer patients. Among them, there were 276 protein data from 166 patients. After the censored data is also deleted, 137×166 lung cancer tumor tissue protein data is obtained. Since lung cancer patients lack normal tissue data, here we select the normal tissue data of breast cancer patients as the control, that is, select 131 tumor tissue proteins of the same type from 166 patients Data and protein data of 131 normal tissues in 45 patients.
[0078] The random forest model is used to encapsulate and filter the protein data of normal tissues of lung cancer patients and the protein data of tumor tissues to select the best subset. In order to select the protein subset with the smallest number of genes and maintain the highest classification accuracy, ten-fold cross-validation was used to evaluate the classifier model, and the protein classification results are shown in Table 4. For the breast cancer data s...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More