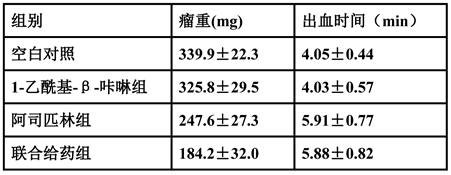
A kind of medicine for treating cervical cancer and its application
A cervical cancer, drug technology, applied in the field of drugs
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030] Example 1 Antitumor Oral Capsules
[0031] Aspirin 2400g
[0032] 1-Acetyl-β-carboline 200g
[0033] Lactose 873.5g
[0034] Povidone K30 78g
[0035] Anhydrous citric acid 69g
[0036] Purified water 700g.
[0037] Add the prescribed amount of povidone K30 and anhydrous citric acid into the purified water of the prescribed amount, and mix them as a binder; take the prescribed amount of aspirin, 1-acetyl-β-carboline and lactose and mix evenly, add the binder to prepare a soft material, Pass through a 18-mesh sieve, dry the wet granules at 50 degrees Celsius and pack into 10,000 capsules.
Embodiment 2
[0038] Example 2 Antitumor Oral Capsules
[0039] Aspirin 3000g
[0040] 1-Acetyl-β-carboline 200g
[0041] Lactose 1100g
[0042] Povidone K30 97g
[0043] Anhydrous citric acid 86g
[0044] Purified water 875g.
[0045] Add the prescribed amount of povidone K30 and anhydrous citric acid into the purified water of the prescribed amount, and mix them as a binder; take the prescribed amount of aspirin, 1-acetyl-β-carboline and lactose and mix evenly, add the binder to prepare a soft material, Pass through a 18-mesh sieve, dry the wet granules at 50 degrees Celsius and pack into 10,000 capsules.
Embodiment 3
[0046] Example 3 Antitumor Oral Capsules
[0047] Aspirin 3400g
[0048] 1-Acetyl-β-carboline 200g
[0049] Lactose 1100g
[0050] Povidone K30 97g
[0051] Anhydrous citric acid 86g
[0052] Purified water 875g.
[0053] Add the prescribed amount of povidone K30 and anhydrous citric acid into the purified water of the prescribed amount, and mix them as a binder; take the prescribed amount of aspirin, 1-acetyl-β-carboline and lactose and mix evenly, add the binder to prepare a soft material, Pass through a 18-mesh sieve, dry the wet granules at 50 degrees Celsius and pack into 10,000 capsules.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com