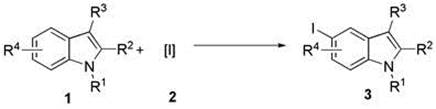
Synthesis method of 5-iodoindole compound
A technology of an iodoindole compound and a synthetic method is applied in the field of organic synthesis and achieves the effects of high reaction efficiency, high yield and simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
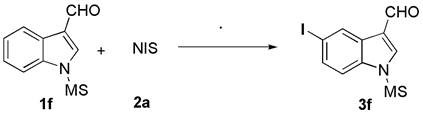
[0018] In a 10 mL one-neck flask, add 0.5 mmol indole-3-carbaldehyde 1a, 0.5 mmol N-iodosuccinimide 2a (NIS), add solvent dichloromethane (3 mL), and add 1 mmol dropwise with stirring BF 3 . Et 2 O, reacted at room temperature for 4 h. After the reaction, extraction was performed, and the solvent was removed by rotary evaporation of the reaction solution under reduced pressure, and the residue was purified by flash silica gel column to obtain the corresponding 5-iodoindole compound 3a in a yield of 78%.
[0019]
[0020] Compound 3a is a white solid, melting point: 185-186 o C; Eluant: ethyl acetate / petroleumether (1:2, R f = 0.30). 1 H NMR (400 MHz, DMSO) δ 12.27 (s, 1H), 9.92 (s, 1H), 8.44 (s, 1H), 8.29 (d, J = 3.1 Hz, 1H), 7.53 (dd, J = 8.5, 1.4 Hz, 1H), 7.37(d, J = 8.5 Hz, 1H); 13 C NMR (101 MHz, DMSO) δ 185.2, 138.9, 136.2, 131.6, 129.2, 126.7, 117.2, 115.0, 86.6; , 1088, 878, 789, 666, 608 cm -1 ; HRMS (ESI) m / z calculated for C 9 H 6 INNaO[M+Na] + :...
Embodiment 2
[0022] Into a 10 mL one-neck flask, add 0.5 mmol indole-3-ethanone 1b, 0.5 mmol iodine, add solvent chloroform (3 mL), and dropwise add 1 mmol BF under stirring 3 . Et 2 O, reacted at room temperature for 4 h. After the reaction was completed, extraction was performed, and the solvent was removed by rotary evaporation of the reaction solution under reduced pressure, and the residue was purified by flash silica gel column to obtain the corresponding 5-iodoindole compound 3b in a yield of 43%.
[0023]
[0024] Compound 3b is a white solid, melting point: 133-134 o C; Eluant: ethyl acetate / petroleumether (1:2, R f = 0.30). 1 H NMR (400 MHz, DMSO) δ 12.08 (s, 1H), 8.52 (d, J =1.6 Hz, 1H), 8.31 (s, 1H), 7.48 (dd, J = 8.5, 1.7 Hz, 1H), 7.33 (d, J = 8.5Hz, 1H), 2.44 (s, 3H); 13 C NMR (101 MHz, DMSO) δ 192.8, 135.8, 135.1, 130.8, 129.7, 127.8, 116.0, 114.6, 86.2, 27.2; , 880, 786, 737, 529 cm -1 ; HRMS (ESI) m / z calculated for C 10 H 8 INNaO[M+Na] + : 307.9543; fo...
Embodiment 3
[0026] In a 10 mL one-neck flask, add 0.5 mmol of indole-3-methyl ester 1c, 0.5 mmol of N-iodosuccinimide 2a, add solvent trichloromethane (3 mL), and dropwise add 1 mmol of trifluorotrifluorocarbon under stirring Acetic acid (TFA) for 4 h at room temperature. After the reaction was completed, extraction was performed, and the solvent was removed by rotary evaporation of the reaction solution under reduced pressure, and the residue was purified by flash silica gel column to obtain the corresponding 5-iodoindole compound 3c in a yield of 61%.
[0027]
[0028] Compound 3c is a yellow solid, melting point: 184-185 o C; Eluant: ethyl acetate / petroleumether (1:2, R f = 0.30). 1 H NMR (400 MHz, DMSO) δ 8.33 (s, 1H), 8.09 (s, 1H), 7.47 (d, J= 8.2 Hz, 1H), 7.35 (d, J = 8.4 Hz, 1H), 3.81 (s, 3H); 13 C NMR (101MHz, DMSO) δ 164.5, 135.6, 133.4, 130.5, 128.8, 128.2, 115.0, 105.7, 85.9, 50.9; IR: 3273, 2923, 1680, 1446, 1357, 119, 4, 79,777 536 cm -1 ;HRMS (ESI) m / z calculated...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More