Homologous recombination mechanism-mediated precise sequence replacement gene editing method and its component structure
A technology of homologous recombination and gene editing, applied in the field of gene editing, can solve the problems of no off-target risk, dependence, high off-target risk, etc., and achieve the effect of increasing the probability of events
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0040] In this example, taking the E. coli gene aroA target segment as an example, how to use the PCRRISR structure and the donor template of the present invention for sequence replacement is described in detail.
[0041] (1) First, prepare a donor template, and the way to obtain the donor template is:
[0042] (1.1) Determine the sequence of the target segment;
[0043] In this example, the E. coli gene aroA was used as the target segment, and its sequence was replaced.
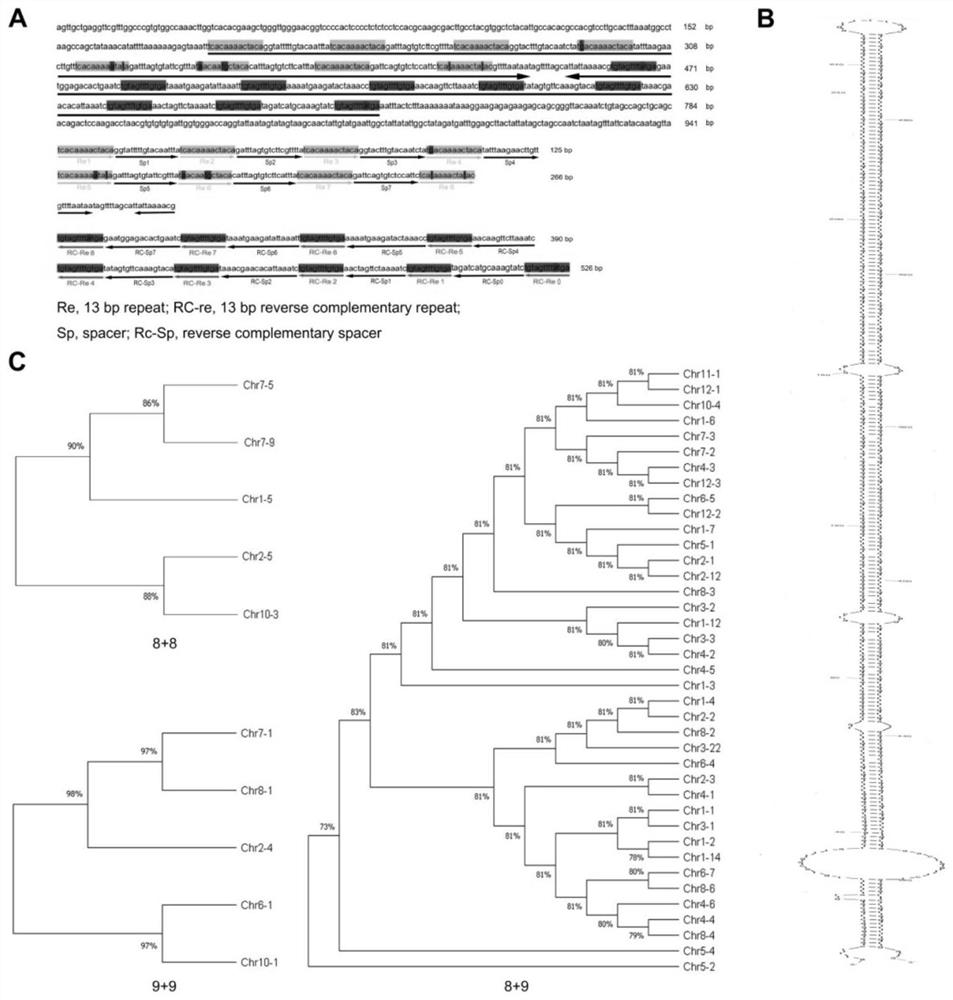
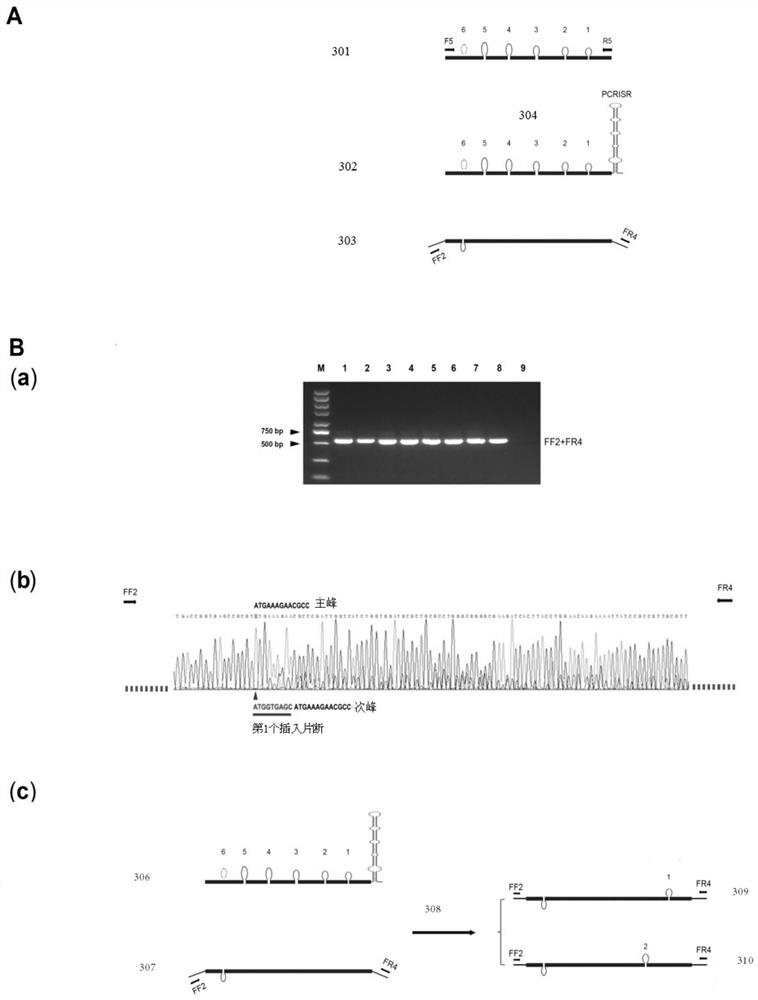
[0044] (1.2) Insert or delete 5-6 donor sites in the target segment sequence to form a donor template. In this example, a MsDFID donor template is used. The MsDFID donor template contains 6 donor sites, 5 of which are DNA fragment insertions, 1 site is DNA fragment deletion, and the length of the homology arm is 60 bp. See image 3 Part (A) of . Preferably, the longest DNA insertion segment is 65 bp, and the longest DNA deletion segment is 35 bp.
[0045] (2) Connect the PCRRISR "hairpin" structure at th...
Embodiment 2
[0060] In this example, the inventors used the NJZL plasmid as the donor template, combined with the PCRRISR "hairpin" structure, to realize the sequence replacement of the target site of the transposon in the E. coli genome.
[0061] (1) First, prepare a donor template, and the way to obtain the donor template is:
[0062] (1.1) Determine the sequence of the target segment;
[0063] In this example, one transposon sequence of Escherichia coli (GenBank No. CP026085.1 (gene product="IS4 family transposase ISVsa5") was used as the target segment to replace its sequence.
[0064] (1.2) Insert or delete several donor sites in the target segment sequence to form a donor template. In this example, the NJZL plasmid donor template is used, and the donor template contains 5 donor sites, see Figure 4 Part (A) of .
[0065] (2) Connect the PCRRISR "issuer" structure to the front end of the donor template
[0066] After inserting multiple donor sites into the target segment sequence,...
Embodiment 3
[0074] In a similar manner to Example 1 and Example 2, in this example, the dctA target segment of the E. coli gene was replaced by sequence, and the replacement results were as follows Figure 5 shown.
[0075] Figure 5 Shown is a diagram of the experimental procedure for the generation of sequence replacement in the dctA target segment of the E. coli gene using a series of vectors comprising a MsDFID donor template fused to a PCRISR construct.
[0076] Figure 5 (A) shows the structure of the donor vector and the electropherogram of the genotyping PCR product obtained by transforming E. coli genomic DNA with these vectors and amplifying it, wherein, in the figure, 501 represents the PCR primer for identifying the genotype of the target segment ; 502 represents the fusion structure of the MsDFID donor template and PCRISR in the donor vector CY7869-6; 503 represents the target segment and its flanking sequence in the E. coli gene dctA; 504 represents the genotype identifica...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More