Preparation method of 6-chloro-2-methyl-2H-indazole-5-amine
A technology of indazole and methyl, which is applied in the field of preparation of 6-chloro-2-methyl-2H-indazol-5-amine, can solve the problems of high price, low reaction yield and high reaction risk, and achieve Improve the convenience of operation, simplify the post-processing operation, and reduce the effect of toxic reagents
Pending Publication Date: 2022-06-10
汉瑞药业(荆门)有限公司
View PDF2 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0005] The yield of the first step of the route reaction is low, and column chromatography is required; the second step of the route reaction needs to use methyl iodide, which is toxic; and NaH is mixed with DMF, which is potentially explosive, and the reaction risk is high; and the reaction Very low yield, requires column chromatography, expensive
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0023]
Embodiment 2
Embodiment 3
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
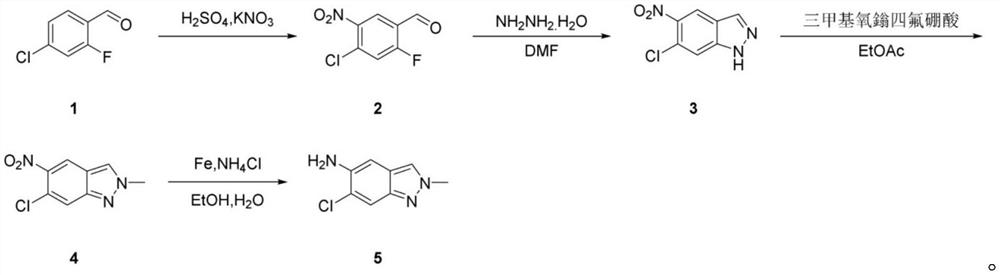
The invention discloses a preparation method of 6-chloro-2-methyl-2H-indazole-5-amine, which sequentially comprises the following steps of: nitrifying 4-chloro-2-fluorobenzaldehyde to obtain 4-chloro-2-fluoro-5-nitrobenzaldehyde, adding potassium nitrate, sulfuric acid and the like in the reaction process, reacting, and filtering to obtain the 6-chloro-2-methyl-2H-indazole-5-amine. The method comprises the following steps: in the presence of hydrazine hydrate, generating 6-chloro-5-nitro-1H-indazole from 4-chloro-2-fluoro-5-nitrobenzaldehyde, then methylating the 6-chloro-5-nitro-1H-indazole by using trimethyloxy onium tetrafluoroboric acid to obtain 6-chloro-2-methyl-5-nitro-2H-indazole, and reducing the 6-chloro-2-methyl-2H-indazole-5-amine to obtain 6-chloro-2-methyl-2H-indazole-5-amine; according to the preparation method disclosed by the invention, toxic reagents and potential explosion risks are reduced, and the operation convenience is improved.
Description
Technical field [0001] The invention belongs to the technical field of pharmaceutical production, and specifically relates to a preparation method of 6-chloro-2-methyl-2H-indazole-5-amine. Background technique [0002] The molecular formula of 6-chloro-2-methyl-2H-indazole-5-amine is Its synthesis is not easy, and the following routes have been reported in relevant literature: [0003] [0004] This route starts from compound A, through ring closure with sodium nitrite, then methylation, and then through reduction to obtain the target compound. [0005] The first step of the reaction in this route has a low yield and requires column chromatography; the second step of the reaction in this route requires the use of methyl iodide, which is toxic; and the mixture of NaH and DMF is potentially explosive and the reaction risk is high; and the reaction The yield is very low, requires column chromatography, and is expensive. Contents of the invention [0006] The object of ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): C07D231/56
CPCC07D231/56
Inventor 柴金柱陈杨李堃杨涛
Owner 汉瑞药业(荆门)有限公司