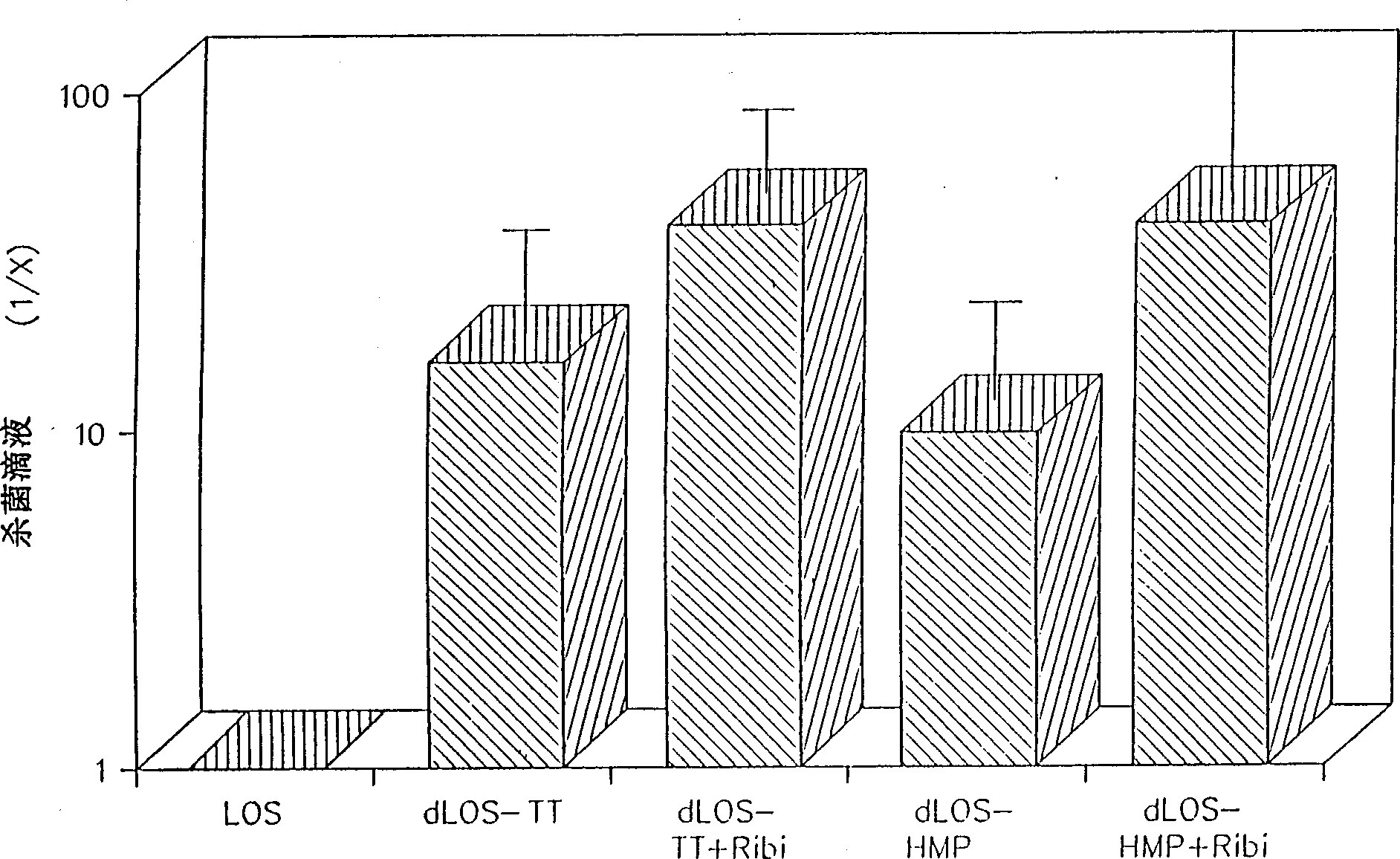Lipooligosaccharide-based vaccine for prevention of i(moraxella) i(branhamella) i (catarrhalis) infections in mammals
A technology based on Moraxella catarrhalis and lipooligosaccharides, applied in the field of conjugated vaccines for the prevention of human Moraxella catarrhalis infection, can solve the problems of weak immunogenicity and other issues
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0026] For the preparation of dLOS or OS conjugates, dLOS can be directly covalently attached to a carrier protein, for example, using a cross-linking reagent such as glutaraldehyde. Preferably, any one of various known methods (for example, see Marburg et al., 1986, J.Am Chem Soc. 108:5282, and U.S. Patent No. 4,882,317; U.S. 5,153,312; U.S. 5,204,098), using The linker compound sequesters the dLOS or OS from the carrier to create a dLOS or OS conjugate. The presence of the linker compound facilitates efficient conjugation of dLOS or OS to the carrier, optimizing the immunogenicity of the conjugate. Linker compounds have side chains of adjustable length and flexibility to sequester carbohydrates and carrier components. Linkers can enhance the translational and rotational properties of the conjugated antigen, thereby increasing the binding pathway of the antibody. Between bifunctional sites, the linker side chains can include various structural features, including heterocycl...
Embodiment 1
[0061] Embodiment 1: Purification and detoxification of Moraxella catarrhalis LOS
[0062]Moraxella catarrhalis (type A) strain ATCC 25238 was used as an exemplary source of purified LOS (Edebrink, P., et al., 1994, Carbohydr. Res. 257:269-284; Masoud, H., et al., 1994, Can. J. Chem. 72:1466-1477). At 37°C, 5% CO 2 The strain was cultured on chocolate agar for 8 hours under the conditions of , and then transferred to 250 ml of 3% tryptic soy broth (TSB) (Difco Laboratories, Detroit, Mich.) in a 500 mL flask. The flask was placed on a constant temperature shaker (Model G-25, New Brunswick Scientific, Co., Edison, NJ) at 37° C. and 110 rpm for overnight culture. The culture was transferred to six 2.8 liter baffled Fernbach flasks each containing 1.4 liters of TSB. Incubate at 37°C and 110 rpm for 24 hours with constant temperature shaking. The cultures were harvested by centrifugation at 15,000 xg for 10 min at 4°C.
[0063] According to the conventional method (basically t...
Embodiment 2
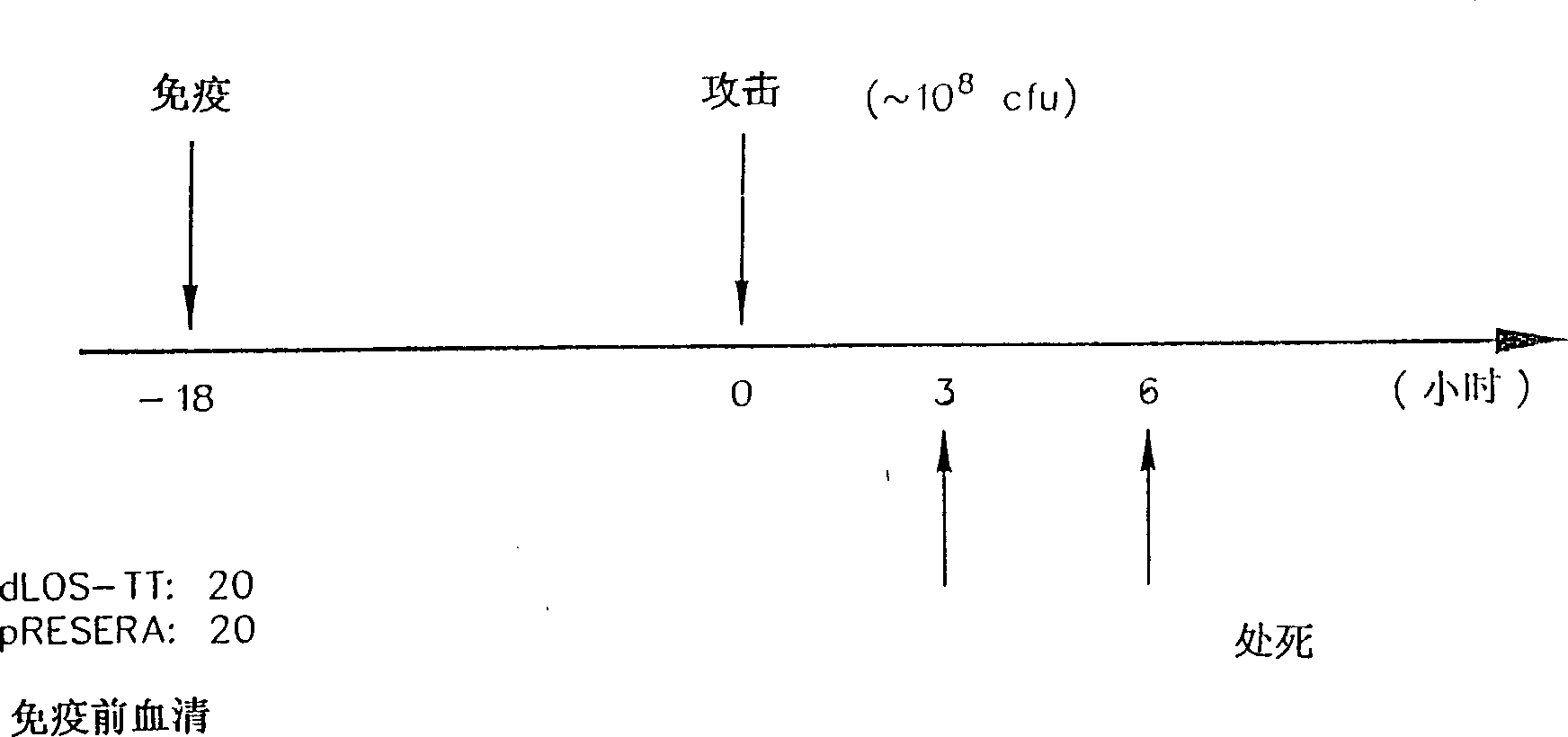
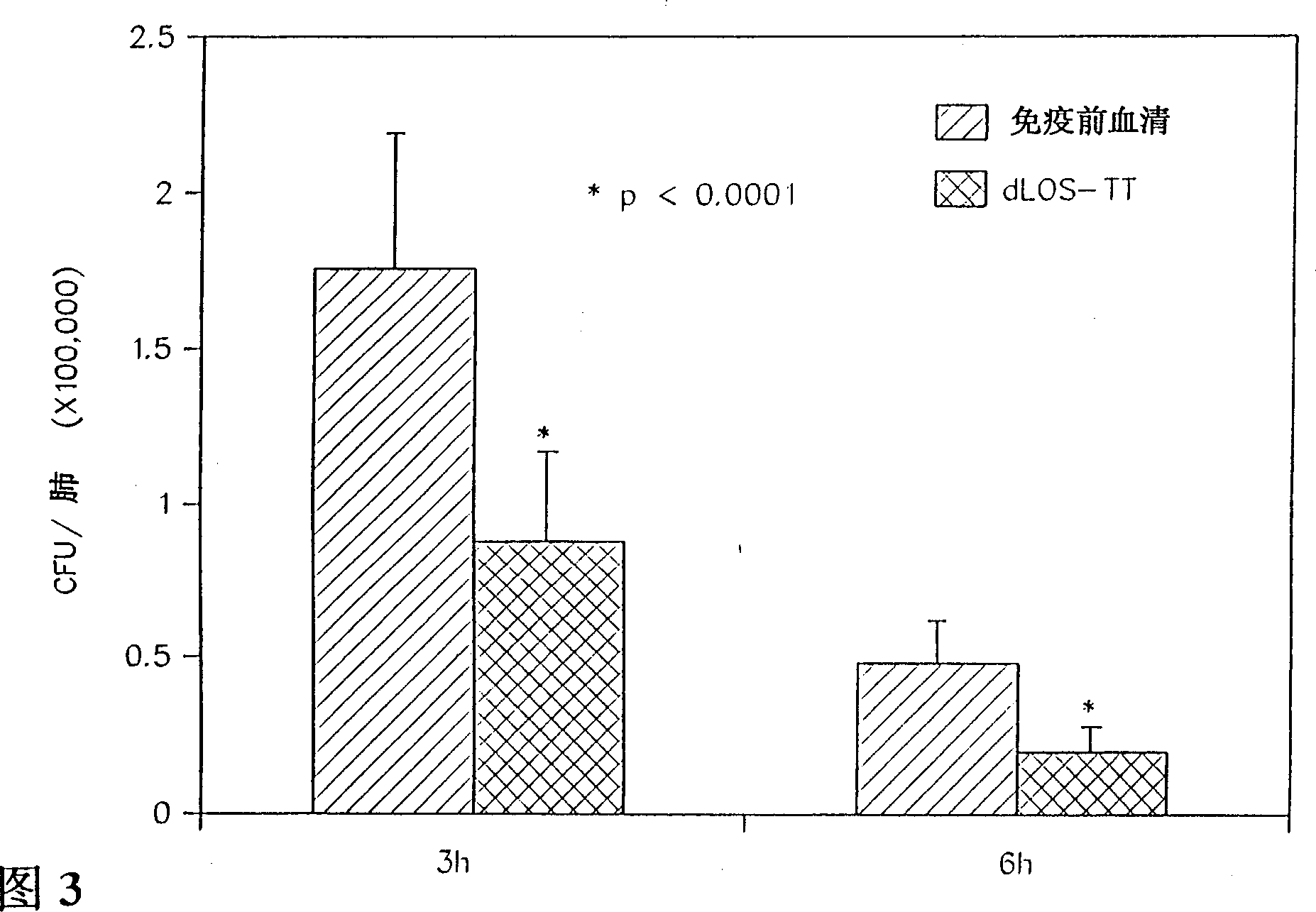
[0066] Example 2: Derivatization of dLOS and conjugation to proteins
[0067] The adipyl hydrazine (AH) derivative of dLOS prepared according to the method in Example 1 was prepared and purified according to the following method. Using 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC) and N-hydroxysulfo-succinimide (sulfo-NHS), di Hydrazide (ADH) is attached to the carboxyl group of the Kdo moiety of dLOS to form an AH-dLOS derivative (Pierce) (Gu, X.X., & C.M. Tsai, 1993, InfecL Immun. 61:1873-1880). Briefly, dLOS (70 mg) was dissolved in 7 mL of 345 mM ADH (the molar ratio of ADH to LOS was about 100:1, based on an estimated dLOS of 3,000 Mr) (Edebrik, P., et al., 1994, Carbohydr. Res. .257:269-284). Add sulfo-NHS to a concentration of 8mM, adjust the pH to 4.8, and add EDC to a concentration of 0.1M. The reaction mixture was stirred and maintained at pH 4.8 for 3 hours. Following the procedure in Example 1, the reaction mixture was adjusted to pH 7.0 an...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More