Methods and compositions for the production of monoclonal antibodies
a monoclonal antibody and composition technology, applied in the field of immunology, can solve the problems of limited development of therapeutic products based on these proteins, no satisfactory method or composition for producing a specifically desired response, and the difficulty of scientists in achieving the so as to improve the uptake of human tnf antigen, improve the efficiency and specificity of the antibody response, and improve the effect of b-cell respons
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Manufacture of Colloidal Gold
[0111] Colloidal gold sols are manufactured using the reaction described by Frens and Horisberger (Frens, G. Nature Phys. Sci. 1972, 241, 20-22, and Horsiberger, M. Biol. Cellulaire. 1979. 36: 253-258). In this reaction ionic gold, in the form of HAuCl4, is reduced to nanoparticles of Au0 by the addition of sodium citrate. Typically, 2.5 ml of a 4% chloroauric acid (in water) solution is added to 1 L of deionized water. The solution is vigorously stirred and heated to a rolling boil. The reduction reaction is initiated by the addition of a 1% sodium citrate solution. The size of the particle is controlled by the amount of citrate added to the reaction. For example, 17, 32, and 64 nm particles are formed by the addition of 40, 15, and 7.5 ml of the citrate solution, respectively. After the addition of citrate, the solution is allowed to boil and mix for an additional 45 minutes. Upon cooling, the sol is filtered through a 0.22 μm sterilization filter and...
example 2
Increasing the Number of Immune Competent B Cells
[0113] To increase the number of immune competent B cells for immunization, MHC class II restricted-surface IgM+ / sIgD+ human B cells are isolated from units of whole blood or buffy coats. Magnetic beads coated with anti-IgM, anti-IgD and anti-CD19 antibodies separate the B cell populations. Treating sIgM+ / sIgD− immature B cells with the cytokine interleukin-7 is used to recruit additional B cells (Sudo, T., Ito, M., Ogawa, Y., Iizuka, M., Kodoma, H., Kunisasa, T., Hayashi, S. C., Ogawa, M., Sakai, K., Nishikawa, S., Nishkawa, S. C. J. Exp. Med. 1989. 170: 333-338). This treatment has been shown to mature these B cells as signaled by the phenotype conversion of sIgM+ / sIgD− B cells to sIgM+ / sIgD+ B cells. These isolated cells are purified to near homogeneity using FACS separation.
[0114] Conjugating TNF to carriers such as KLH or thyroglobulin (see discussion below) enhances the antigenicity of human TNF. TNF:KLH antigen is bound to th...
example 3
Differentiation of the Primary Antibody Response
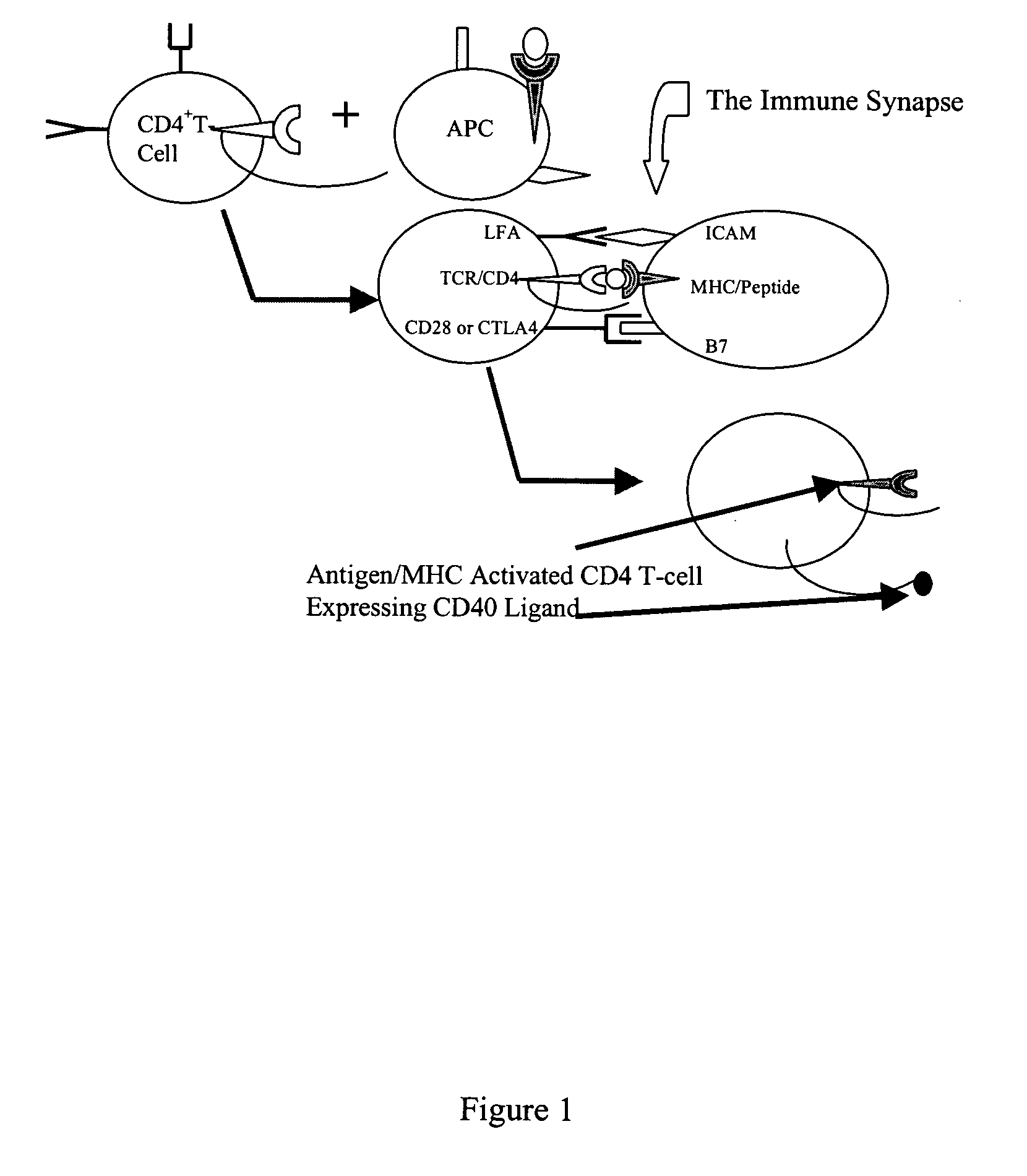
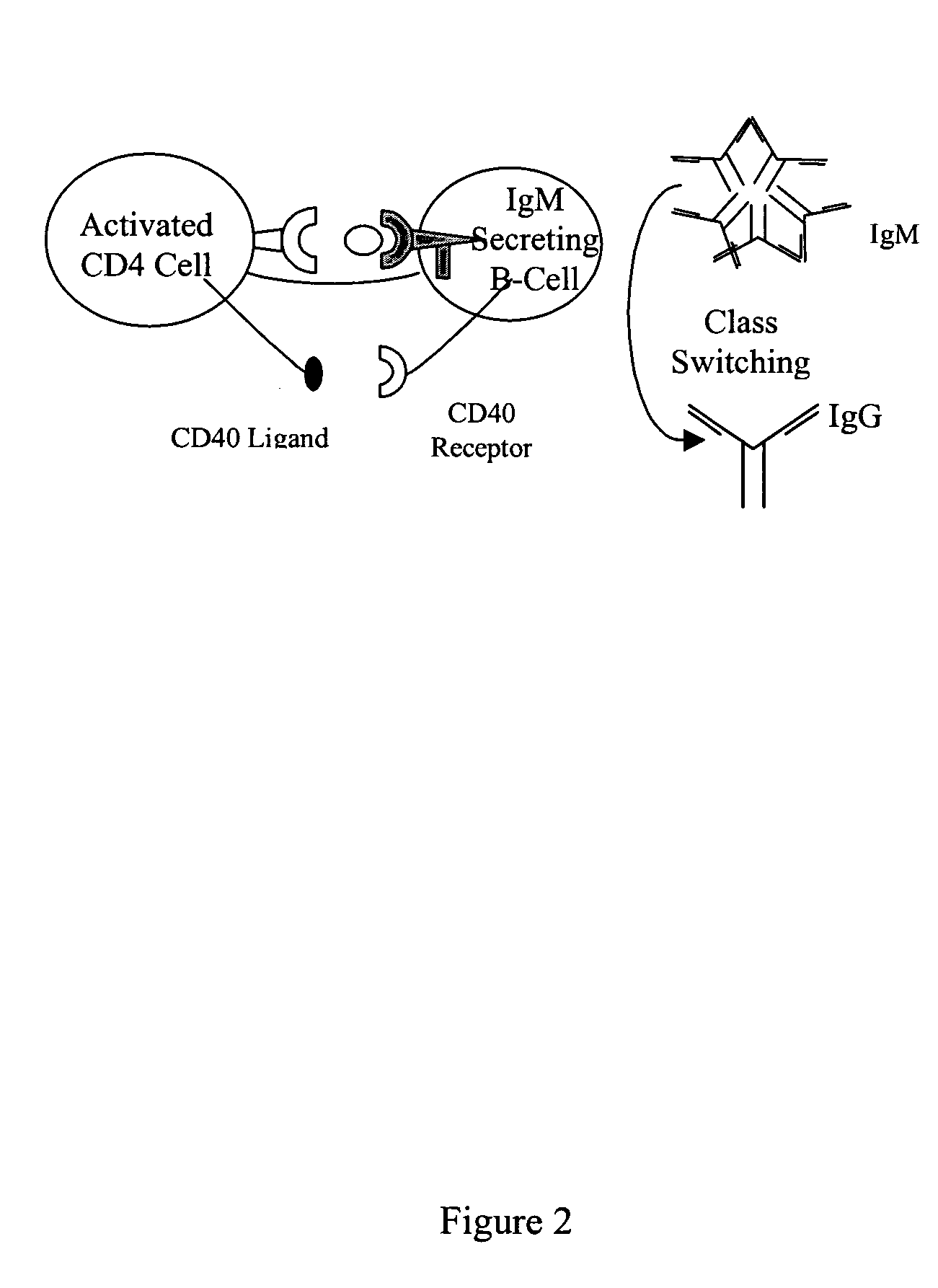
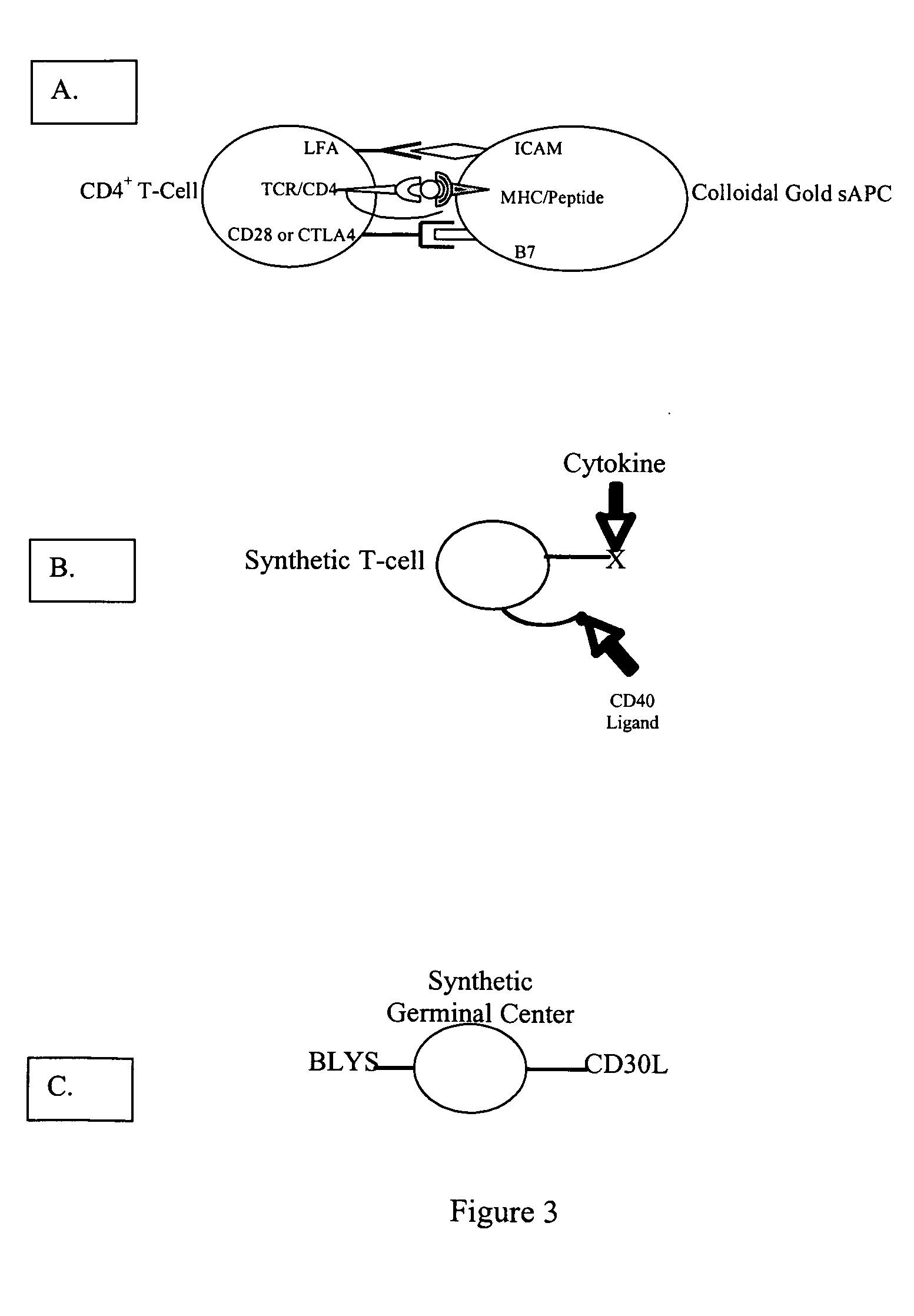
[0115] Critical to the production of a therapeutic antibody is the process of class switching. The primary antibody response from immunized human B cells results in the secretion of IgM antibodies. A second class of lymphoid cells, known as antigen presenting cells (APCs), also internalizes the antigen. Once internalized these cells process the protein antigen into fragments, which are then expressed on the cell's surface bound to one of two major histocompatibility complexes (MHCs).
[0116] Depending on the microenvironment, APCs expressing antigen bound to class II MHC molecules activate one of two subsets of CD4+ T cells. These cells, also known as helper T cells, perform the necessary accessory functions to facilitate the cellular or the humoral (antibody) immune response. TH1 CD4+ cells facilitate the cellular immune response, while the TH2 subset of CD4+ cells interact with IgM secreting B cells to initiate the process of class s...
PUM
| Property | Measurement | Unit |
|---|---|---|
| flow rate | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| concentrations | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More