Glucan-based vaccines
a glucan-based, vaccine technology, applied in the field of vaccines, can solve the problems low protective efficacy, and ineffective whole-cell vaccines, and achieve the effect of low protective efficacy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
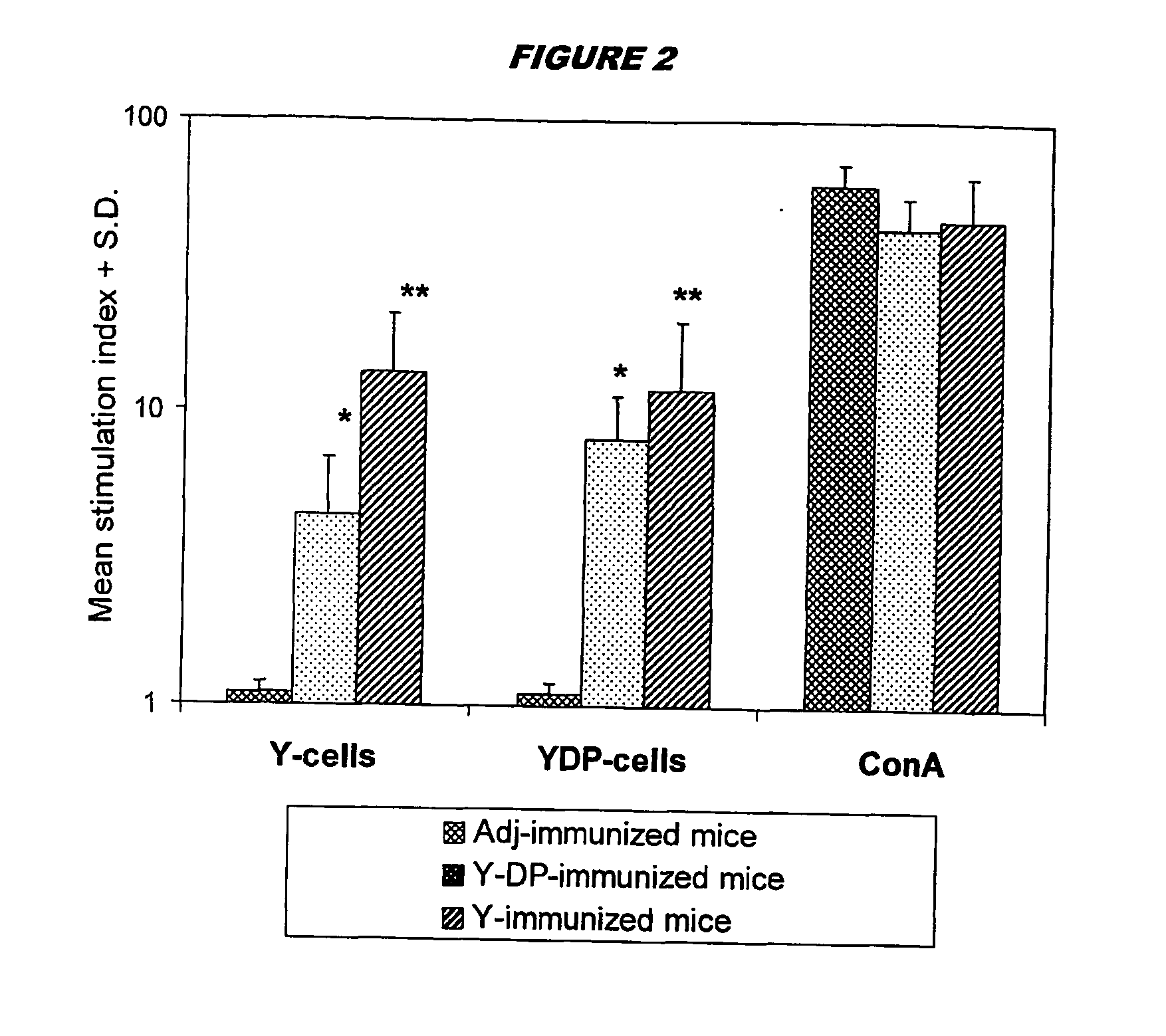
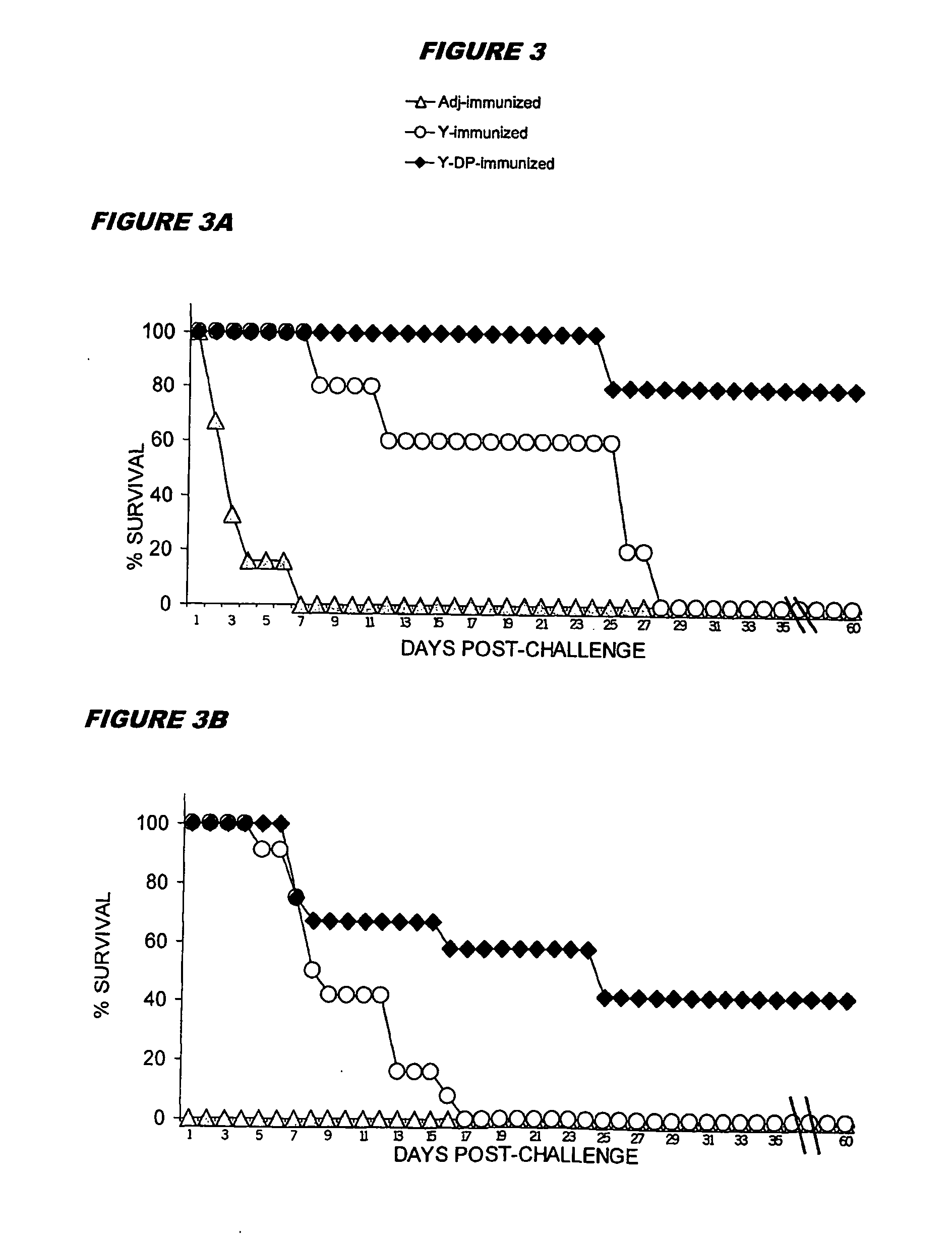
Preparation of Mannoprotein-Depleted Yeast Cells
[0133]C. albicans strain BP, serotype A, from the type collection of the Istituto Superiore di Sanità (Rome, Italy), was routinely maintained on Sabouraud agar slants. For all experiments, fungus was cultured in the yeast form in liquid Winge medium at 28° C., washed twice in saline, counted in a haemocytometerer, and resuspended at the desired concentration in sterile saline.
[0134] For the preparation of normal cells (‘Y cells’) yeast cells suspensions (108 cells / ml) were inactivated at 80° C. for 30 min, washed and stored at 4° C. for no more than a week.
[0135] To prepare mannoprotein-depleted cells (‘YDP cells’), heat-inactivated Y cells as above (108 / ml) were treated with 50 mM DTT in 5 mM EDTANa2 (1 hour, 37° C.). 500 μg / ml Proteinase K (Sigma) was added to the digestion mixture and the cells were treated for one further hour at 37° C. The fungal cells were extensively washed with saline to remove enzyme, resuspended in saline...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Atomic weight | aaaaa | aaaaa |
| Density | aaaaa | aaaaa |
| Density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More