2'-deoxy-2'alkylnucleotide containing nucleic acid
a technology of alkylnucleotide and ribozyme, which is applied in the direction of organic chemistry, peptide/protein ingredients, genetic material ingredients, etc., can solve the problems of limiting the potential use of rnasep-like rna in therapeutics, destroying the ability to direct synthesis of encoded proteins, and reducing the cost of treatment. , the therapeutic cost of such molecules is prohibitive, and the degree of specificity high
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
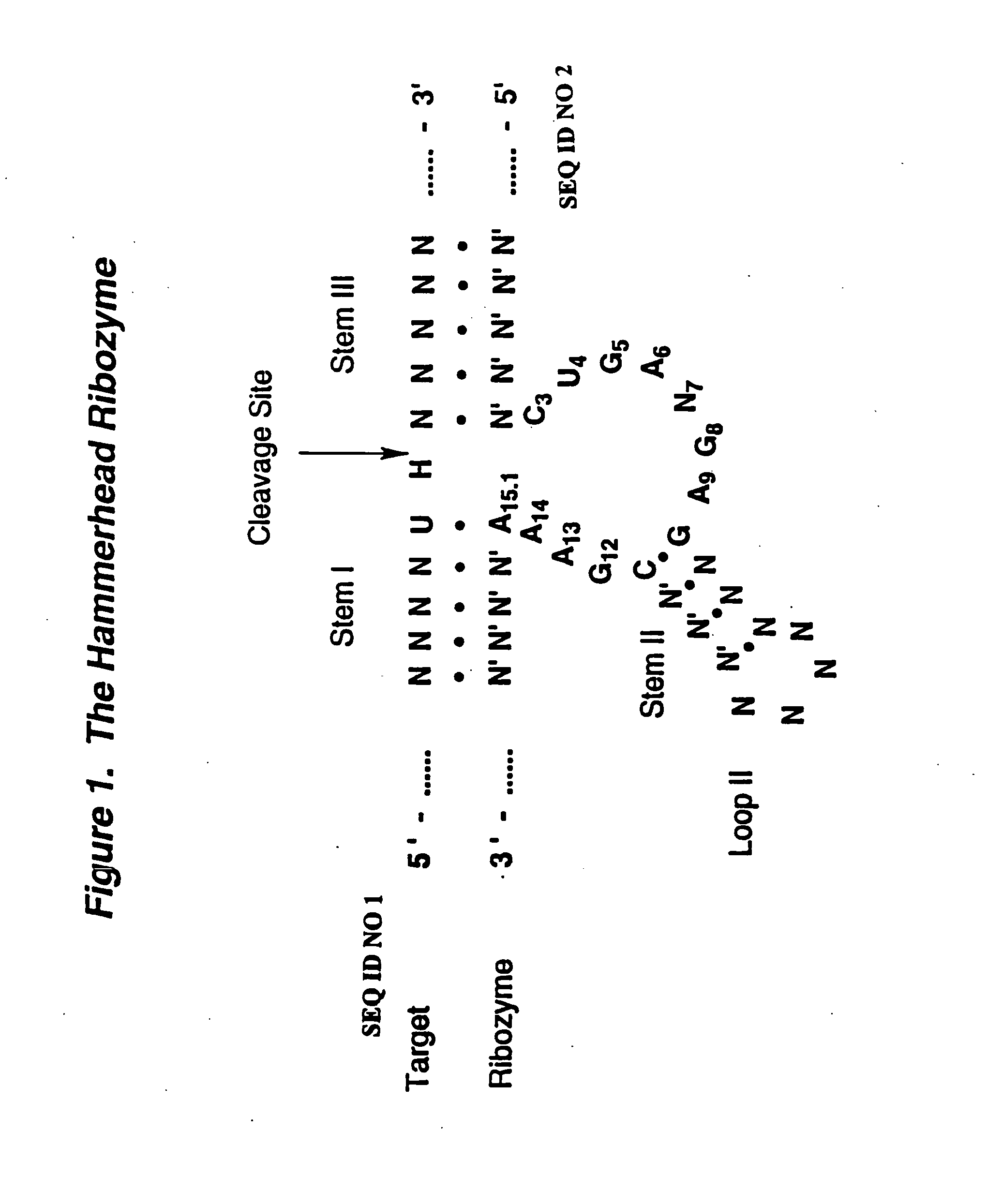
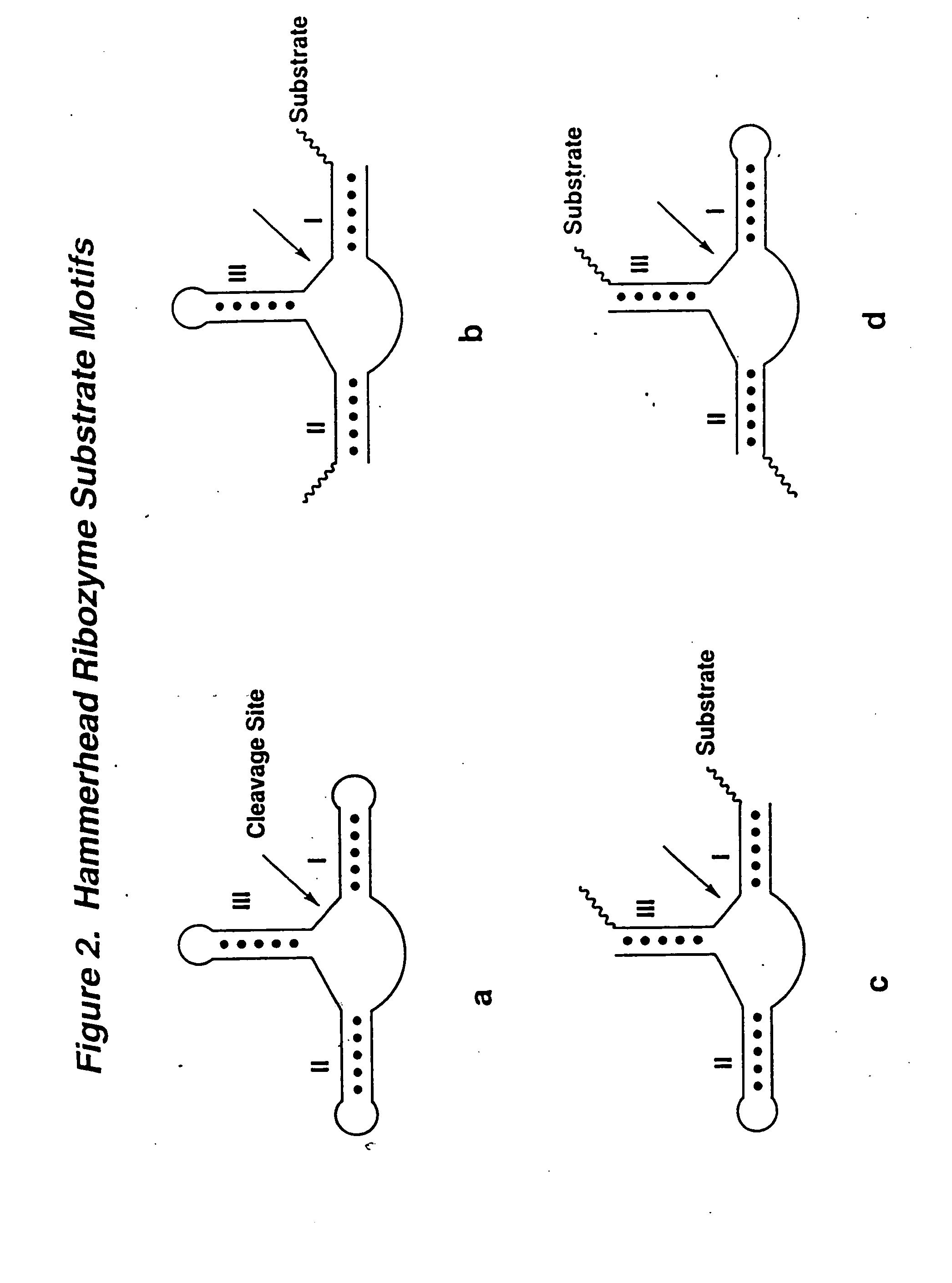
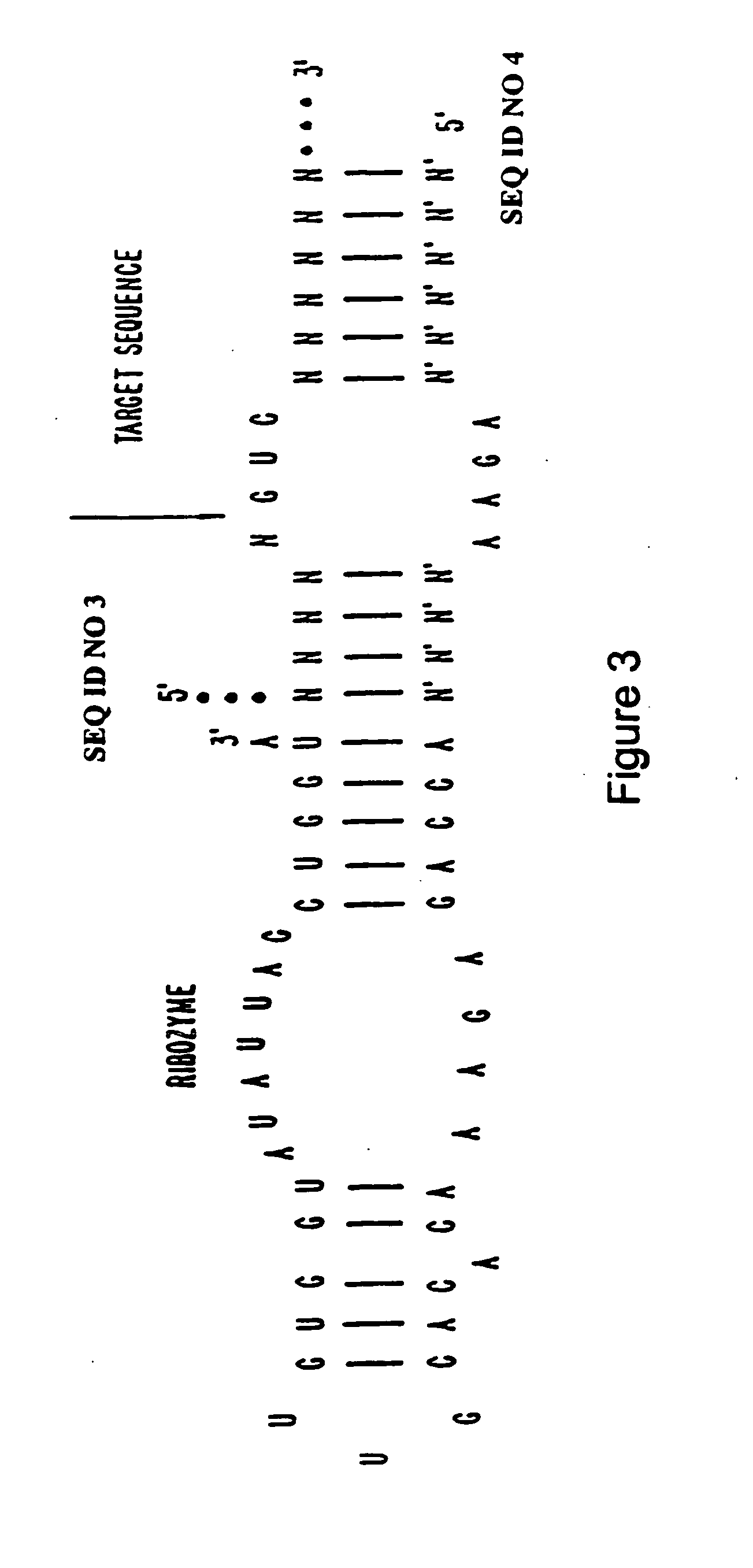
Synthesis of Hammerhead Ribozymes Containing 2′-Deoxy-2′-Alkylnucleotides & Other 2′-Modified Nucleotides
[0044] The method of synthesis used generally follows the procedure for normal RNA synthesis as described in Usman, N.; Ogilvie, K. K.; Jiang M.-Y.; Cedergren, R. J. J. Am. Chem. Soc. 1987, 109, 7845-7854 and in Scaringe, S. A.; Franklyn, C.; Usman, N. Nucleic Acids Res. 1990, 18, 5433-5441 and makes use of common nucleic acid protecting and coupling groups, such as dimethoxytrityl at the 5′-end, and phosphoramidites at the 3′-end (compounds 10, 12, 17, 22, 31, 18, 26, 32, 36 and 38). Other 2′-modified phosphoramidites were prepared according to: 3 & 4, Eckstein et al. International Publication No. WO 92 / 07065; and 5 Kois et al. Nucleosides &Nucleotides 1993, 12, 1093-1109. The average stepwise coupling yields were ˜98%. The 2′-substituted phosphoramidites were incorporated into hammerhead ribozymes as shown in FIG. 5. However, these 2′-alkyl substituted phosphoramidites may be ...
example 2
[0045] Purified 5′-end labeled RNA substrates (15-25-mers) and purified 5′-end labeled ribozymes (˜36-mers) were both heated to 95° C., quenched on ice and equilibrated at 37° C., separately. Ribozyme stock solutions were 1 mM, 200 nM, 40 nM or 8 nM and the final substrate RNA concentrations were ˜1 nM. Total reaction volumes were 50 mL. The assay buffer was 50 mM Tris-Cl, pH 7.5 and 10 mM MgCl2. Reactions were initiated by mixing substrate and ribozyme solutions at t=0. Aliquots of 5 mL were removed at time points of 1, 5, 15, 30, 60 and 120 m. Each time point was quenched in formamide loading buffer and loaded onto a 15% denaturing polyacrylamide gel for analysis. Quantitative analyses were performed using a phosphorimager (Molecular Dynamics).
example 3
Stability Assay
[0046] 500 pmol of gel-purified 5′-end-labeled ribozymes were precipitated in ethanol and pelleted by centrifugation. Each pellet was resuspended in 20 mL of appropriate fluid (human serum, human plasma, human synovial fluid or fetal bovine serum) by vortexing for 20 s at room temperature. The samples were placed into a 37° C. incubator and 2 mL aliquots were withdrawn after incubation for 0, 15, 30, 45, 60, 120, 240 and 480 m. Aliquots were added to 20 mL of a solution containing 95% formamide and 0.5×TBE (50 mM Tris, 50 mM borate, 1 mM EDTA) to quench further nuclease activity and the samples were frozen until loading onto gels. Ribozymes were size-fractionated by electrophoresis in 20% acrylamide / 8M urea gels. The amount of intact ribozyme at each time point was quantified by scanning the bands with a phosphorimager (Molecular Dynamics) and the half-life of each ribozyme in the fluids was determined by plotting the percent intact ribozyme vs the time of incubation...
PUM
| Property | Measurement | Unit |
|---|---|---|
| volumes | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com