Method for promoting bone formation
a bone-specific alkaline phosphatase and bone-specific technology, applied in the field of bone-specific alkaline phosphatase, can solve the problems of bone loss, inability to achieve the effect of such treatment, and the balance of constructive and destructive processes is tipped from equilibrium, so as to promote bone growth, prevent bone loss, and promote bone growth
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Clinical Testing
Patient Studies
[0060] Protocols 1-7 for use of RenaGel® (Geltex Pharmaceuticals, Inc., Waltham, Mass.) in each of the haemodialysis patient studies are provided in the following references, respectively, the teachings of which are incorporated herein by reference in their entireties.
Protocol 1:
[0061] Chertow, G. M., Burke, S. K., Lazarus, J. M., Stenzel, K. H., Wombolt, D., Goldberg, D., Bonventre, J. V., and Slatopolsky, E., “Poly(allylamine hydrochloride) (RenaGel®): a noncalcemic phosphate binder for the treatment of hyperphosphatemia in chronic renal failure,”Am. J. Kid. Dis. 29: 66-71 (1997).
Protocol 2: [0062] Goldberg, D. I., Dillon, M. A., Slatopolsky, E. A., Garrett, B., Gray, J. R., Marbury, T., Weinberg, M., Wombolt, D., and Burke, S. K., “Effect of RenaGel, a non-absorbed, calcium- and aluminum-free phosphate binder, on serum phosphorus, calcium, and intact parathyroid hormone in end-stage renal disease patients,”Nephrol. Dial. Transplant. 13: 2303...
example 2
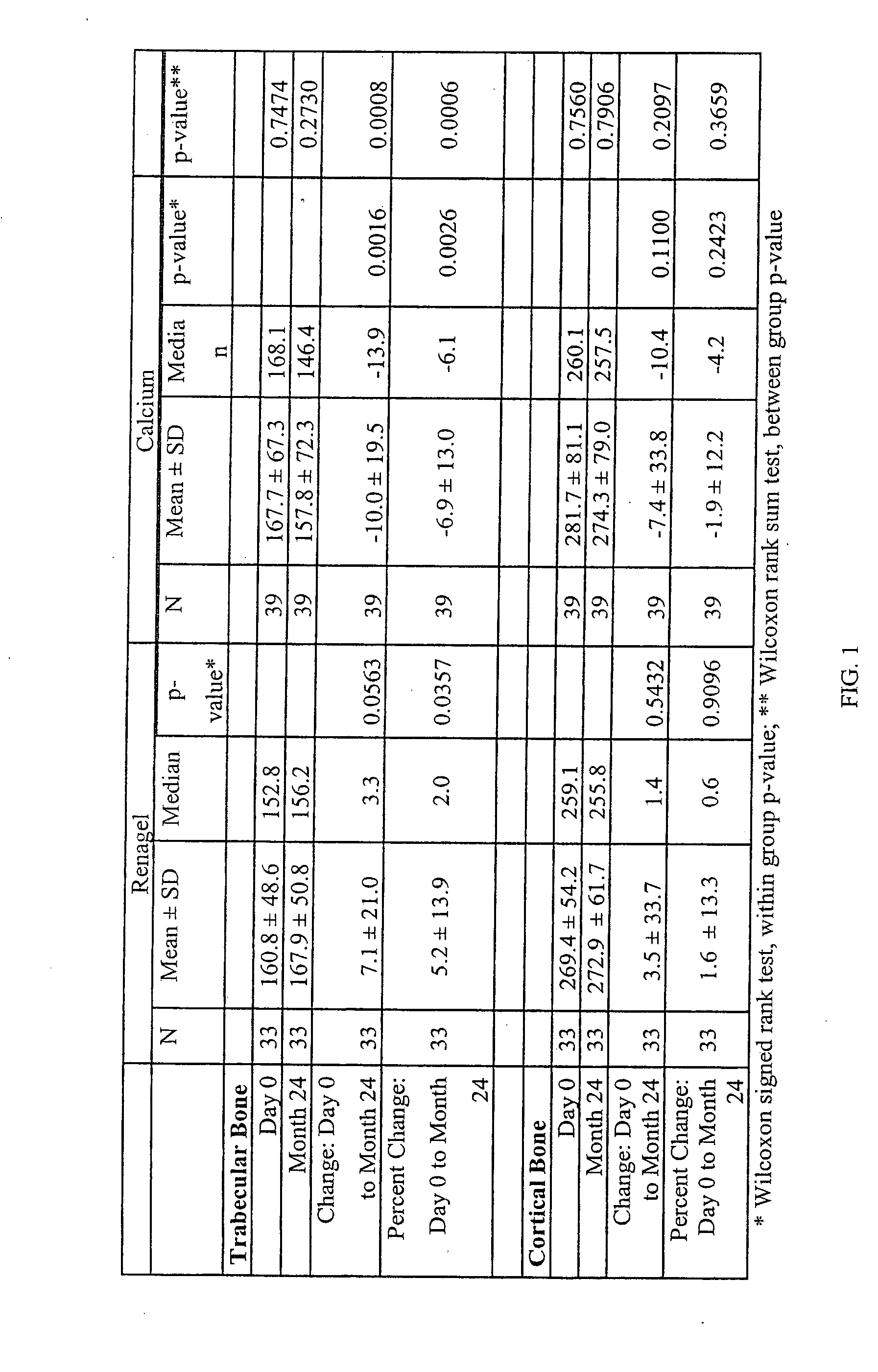
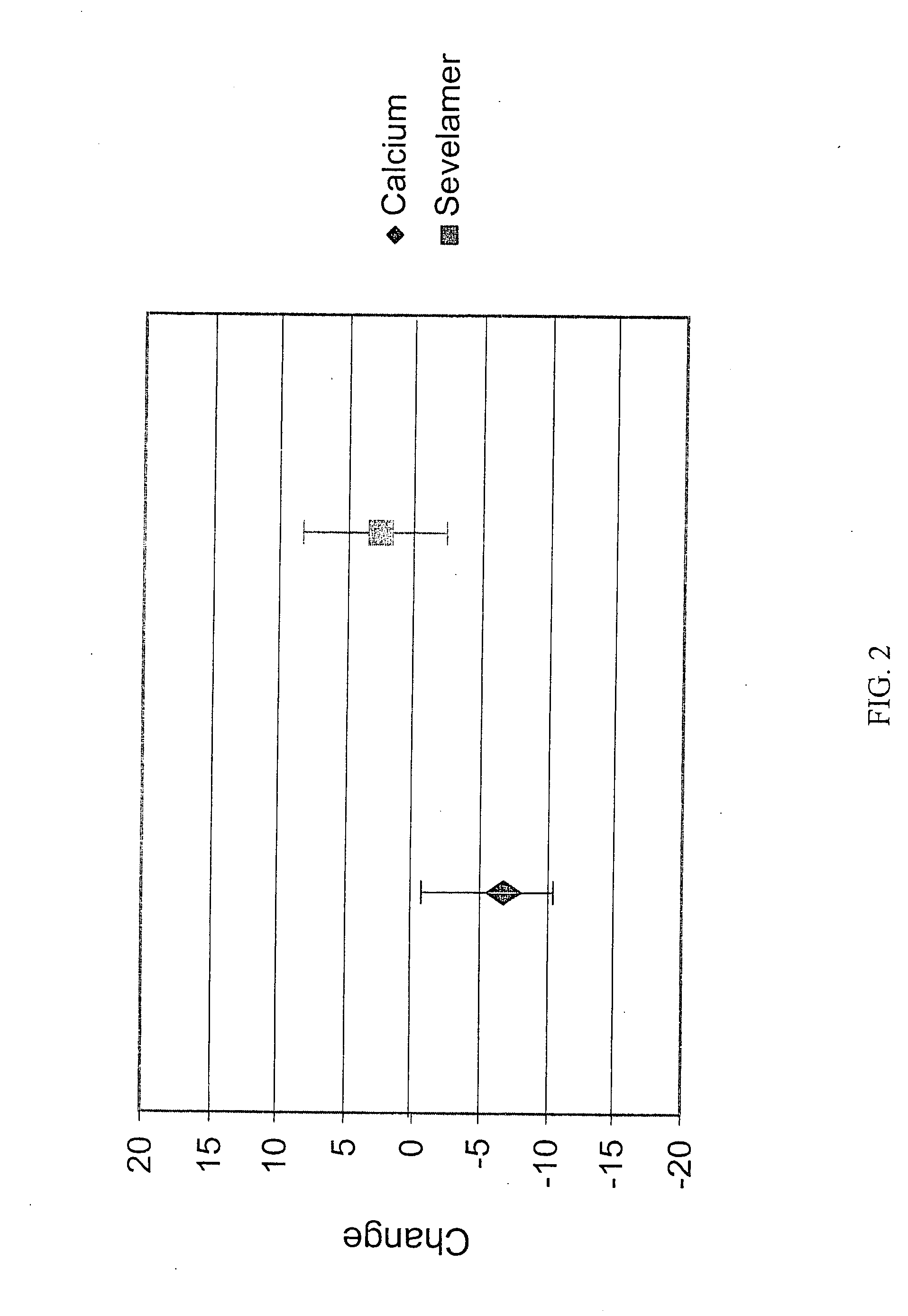
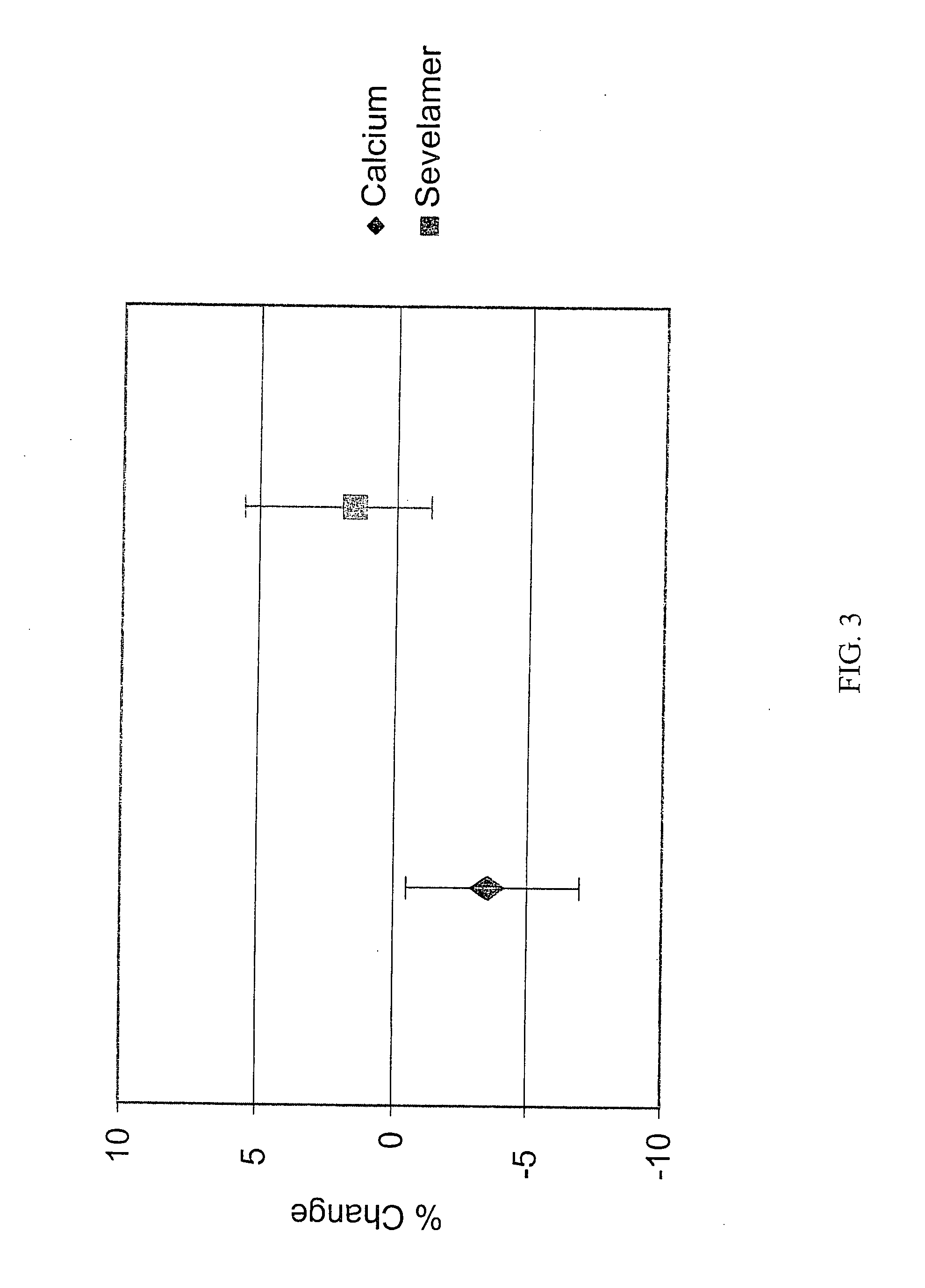
[0075] Upon finding that there were effects on bone in the patients studied in Example 1, an additional analysis was conducted in the larger group of patients from the original study who had a baseline and 12 month bone density scan.
Results
[0076] The study groups (calcium and sevelamer) were well balanced by randomization. At baseline, there were no significant differences between groups for any biochemical parameter measured except for slightly higher HDL cholesterol in the group randomized to calcium compared (calcium 50±17 mg / dL vs. sevelamer 44±15 mg / dL, p=0.04).
[0077] Following the 12 months of treatment, the levels a number of biochemical markers were assayed in the patient groups. Serum phosphorus and calcium-phosphorus product were not significantly different between treatment groups. The group treated with sevelamer had on average higher iPTH, 1-84 PTH, alkaline phosphatase, bone specific alkaline phosphatase, osteocalcin, and HDL cholesterol. The sevelamer group had lo...
PUM
| Property | Measurement | Unit |
|---|---|---|
| aliphatic | aaaaa | aaaaa |
| strength | aaaaa | aaaaa |
| density | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More