Method for thermo-chemical energy storage
Inactive Publication Date: 2019-06-13
VIENNA UNIVERSITY OF TECHNOLOGY +1
View PDF0 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
The present invention is about a new system for producing ammine complexes of transition metal salts. Unlike previous systems, this invention only allows the ammine complex formation reaction to occur, rather than two parallel reactions that complement each other. This means that the entire enthalpy of formation of the ammine complex is recovered during the reaction, which is very high. However, the invention solves this problem by applying the metal salts onto a carrier simultaneously with the formation reaction, which prevents melting and agglutination of the salts. The use of a particulate carrier material with a specific grain size makes it easier to handle and react the materials. This results in increased working life of the carrier material and more efficient energy utilization.
Problems solved by technology
They are still at a very early stage in their development, though, and for most of them a practical or economic application is still years away.
In most known cases, however, the energy storage density of the systems named above is rather low, and the corrosivity of some of the salts used often presents a problem with respect to the equipment.
Additionally, transport and storage of the metal salts used cause problems, because the temperatures reached during the exothermic reaction are frequently near or even above the melting point of the salts / complexes, so that at least a part of the respective salts will melt, which leads to agglutination.
Furthermore, for all known systems for thermo-chemical energy storage, the quality of the thermal storage material used decreases as the number of cycles increases, so that it either has to be exchanged or, in some cases, cleaned expensively.
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
synthesis example 1
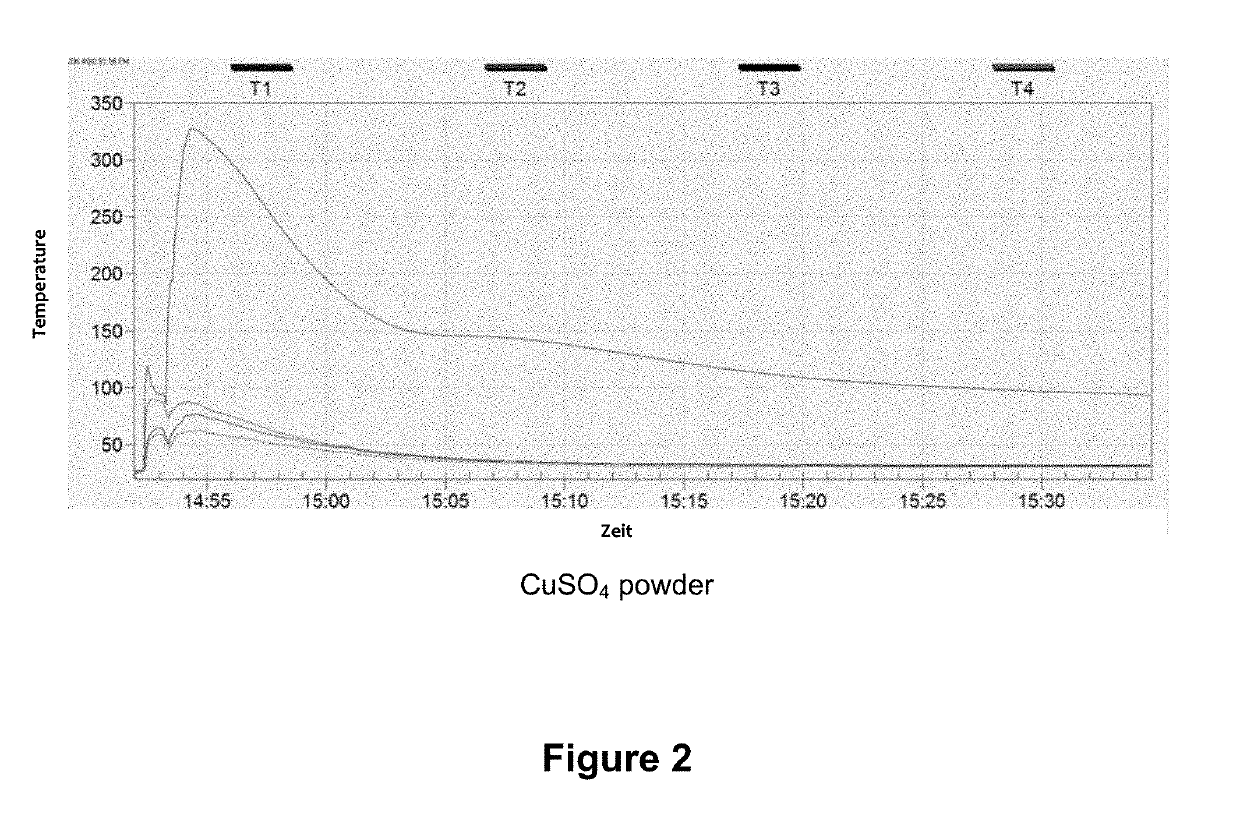
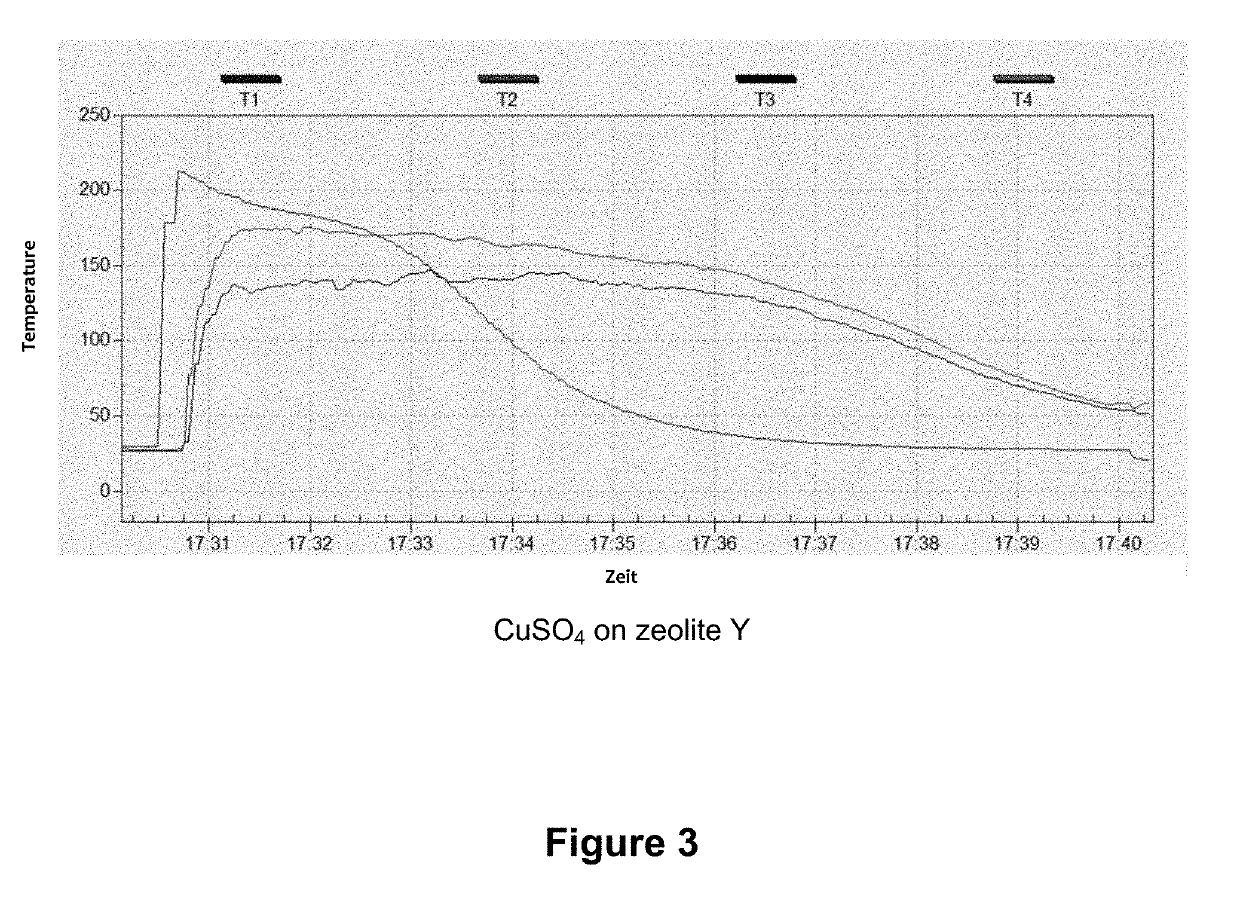
[0031]In a typical loading experiment, 50 g of zeolite Y, which had previously been dried for 6 h at 400° C., were immersed into a saturated CuSO4 solution for 2 h and thus soaked therewith. The bluish product was then separated, flushed with 200 ml of deionized water, and dried for 12 h at 60° C. and thereafter for 6 h at 350° C. A CuSO4 content of 39.7 wt % was determined using XRF analysis.
synthesis example 2
[0032]In a manner analogous to Synthesis Example 1, zeolite Y was soaked with a saturated CuCl2 solution. A CuCl2 content of 44.3 wt % was determined using XRF analysis.
synthesis example 3
[0033]In a manner analogous to Synthesis Example 1, zeolite Y was soaked with a saturated CdCl2 solution. A CdCl2 content of 59.5 wt % was determined using XRF analysis.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
A method for thermo-chemical energy storage involves carrying out reversible chemical reactions for the storage of heat energy in the form of chemical energy in one or more chemical compounds for later re-release in the form of heat energy using chemical equilibrium reactions. Equilibrium reactions of ammine complexes of transition metal salts are carried out for the storage and re-release of the energy. Specifically, ammine complexes of transition metal salts are formed and decomposed according to the following reversible total reactions: [Me(NH3)n]X+ΔHR⇄MeX+n NH3, wherein Me represents at least one transition metal ion and X represents one or more counterion(s) in a quantity sufficient for charge equalizing the complex, according to the valence thereof and that of the transition metal ion. Also, one or more transition metal salt(s), carried on a carrier material that is inert with regard to the reaction, is / are used.
Description
[0001]The present invention relates to a method for thermo-chemical energy storage by carrying out endothermic chemical reactions for the storage of heat energy in the form of chemical energy in one or more chemical compounds for later re-release in the form of heat energy by carrying out chemical equilibrium reactions of ammine complexes of transition metal salts.PRIOR ART[0002]Thermo-chemical energy storage, i.e. storage of heat energy in the form of chemical energy, is a method of energy storage by cycling at least one chemical compound between the states of at least one reversible equilibrium reaction, said method having been known for decades, but only been subject to more intense research in the last few years. For example, U.S. Pat. No. 4,365,475 discloses the combination of two equilibrium reactions for the purpose of thermo-chemical energy storage, namely the alternating reversible endothermic formation of two ammine complexes, CaCl2.8NH3 and ZnCl2.NH3.[0003]In general, the...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): F28D20/00
CPCF28D20/003C09K5/16Y02E60/14
Inventor MUELLER, DANNYKNOLL, CHRISTIANWEINBERGER, PETERWERNER, ANDREAS
Owner VIENNA UNIVERSITY OF TECHNOLOGY