Stentless aortic valve replacement with high radial strength
a radial strength, aortic valve technology, applied in the field of stentless aortic valve replacement with high radial strength, can solve the problems of cardiac insufficiency, potentially fatal for patients, and operation becomes particularly difficult, and achieves the effects of high radial strength, improved quality of life, and minimal invasiveness
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0043]The present invention provides a percutaneous valve replacement with high radial strength.
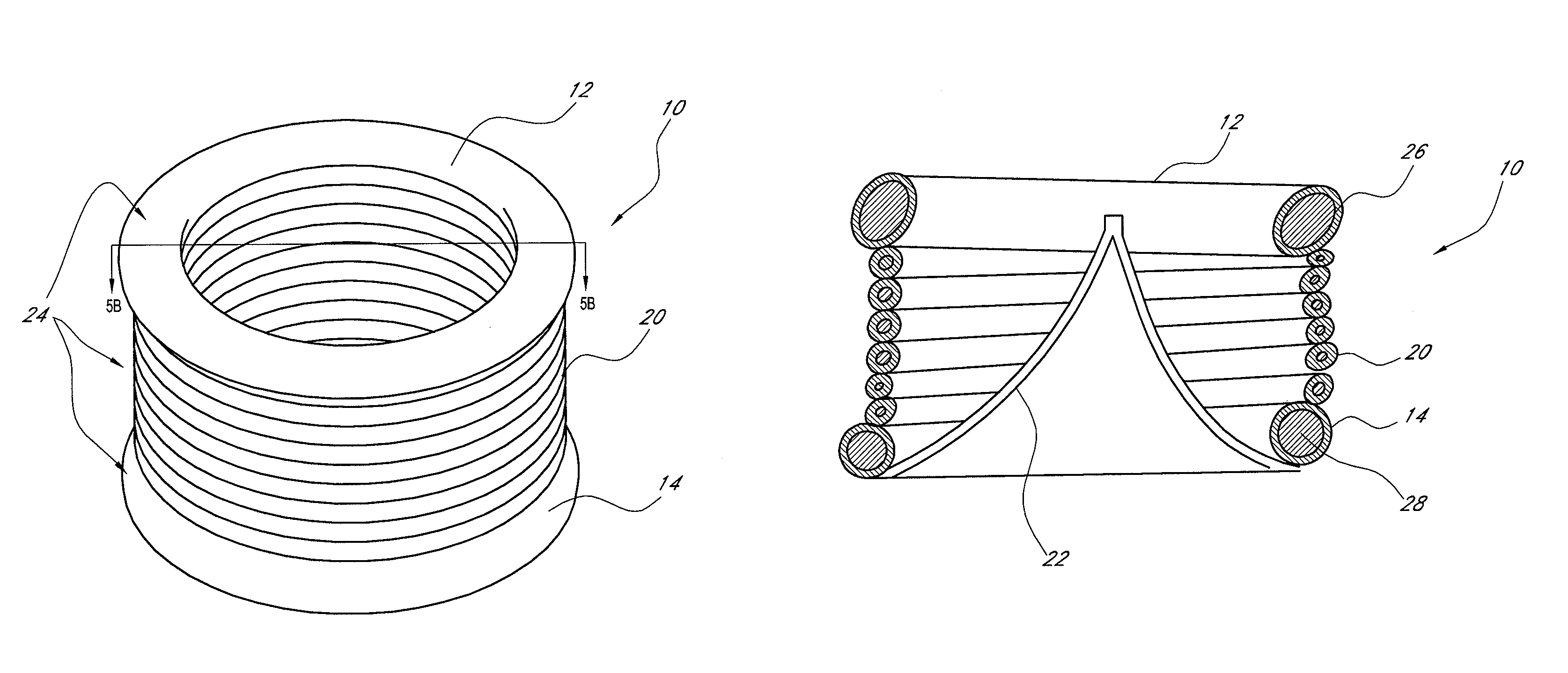
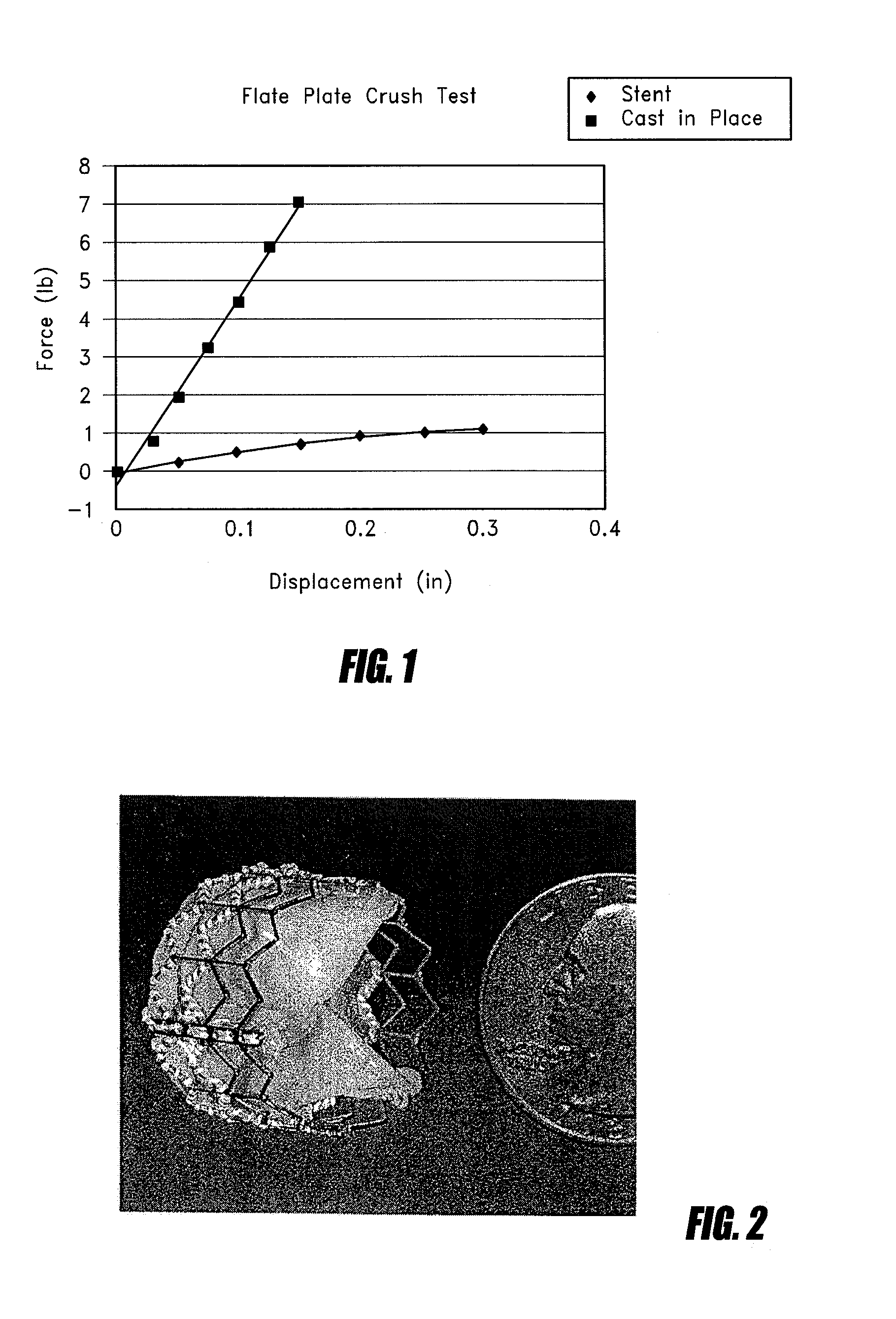
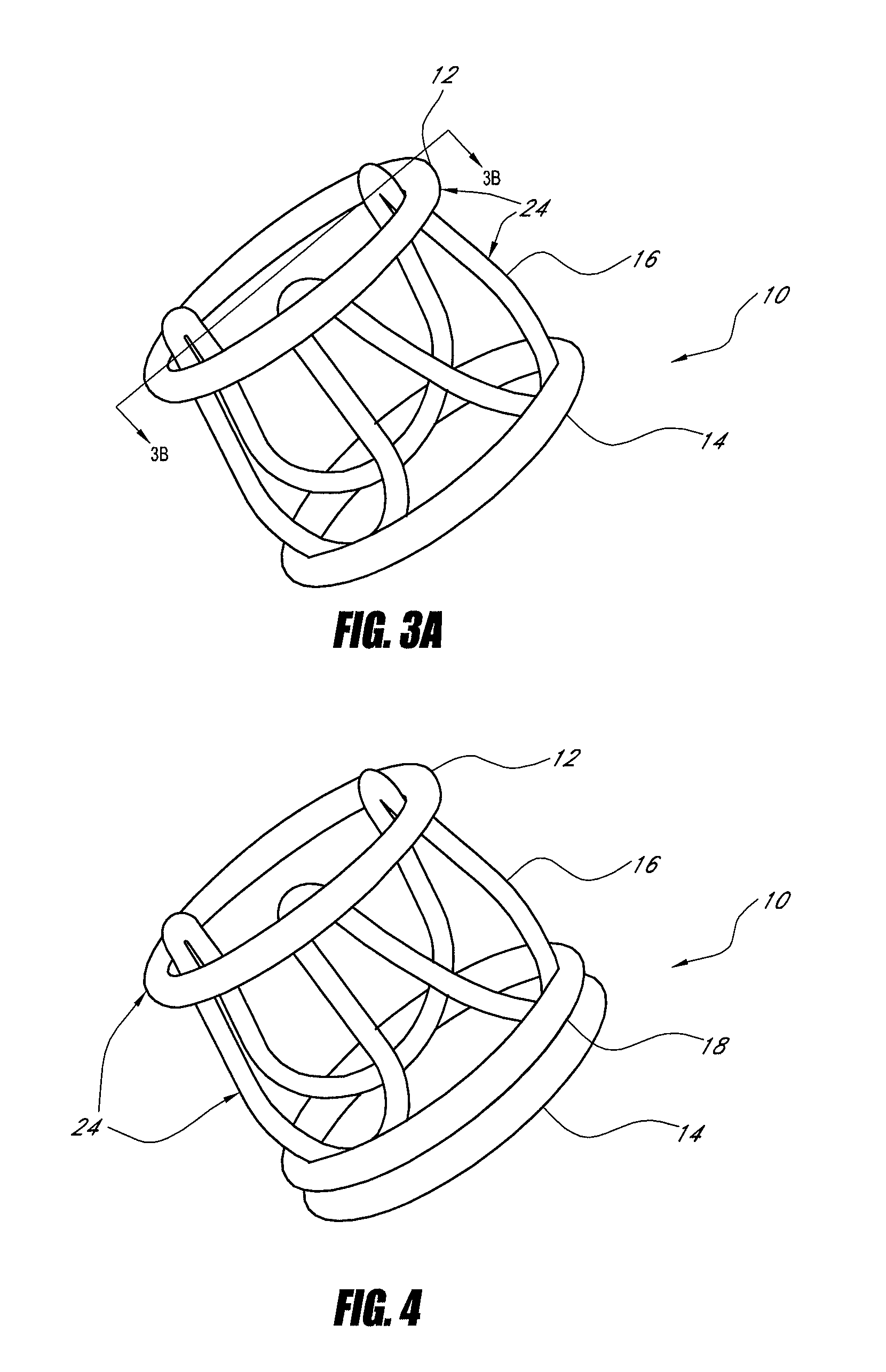
[0044]One current method for implanting a tissue valve percutaneously includes a balloon expandable or self-expanding stent with a tissue valve attached as described in Andersen U.S. Pat. No. 6,168,614. See FIG. 2. Another method to implant a tissue valve percutaneously is described in U.S. Pat. No. 5,554,185 (Block), the disclosure of which is incorporated in its entirety herein by reference. One key feature in any valve apparatus is the ability to withstand forces generated by the closure of the valve at the commissural supports. In surgical valves these are seen as posts or pillars rising from the base of the device. General construction often includes a metallic frame encompassed with silicone and wrapped with Dacron. This frame will withstand the cyclical loading seen under normal conditions of operation in a heart valve. Since surgical valves are installed under direct visualization...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More