Citric acid alidenafil crystal form D and preparation method and usage thereof
A citric acid, amorphous technology, applied in the field of citrate aldenafil crystalline form D and its preparation and use, can solve the problems that do not involve the citrate aldenafil crystalline form and its preparation method, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
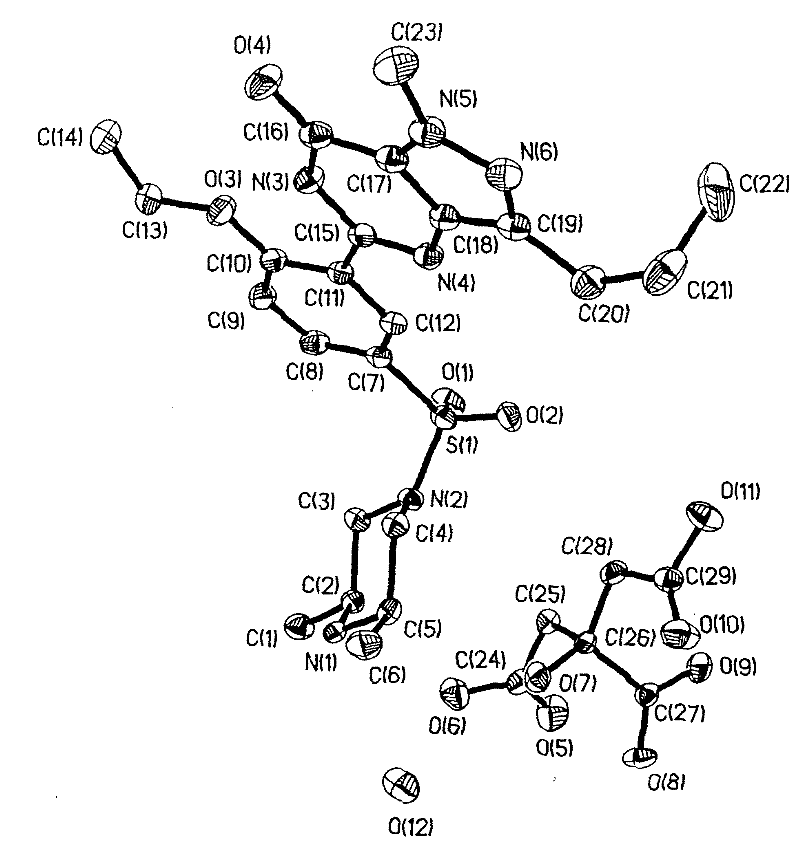
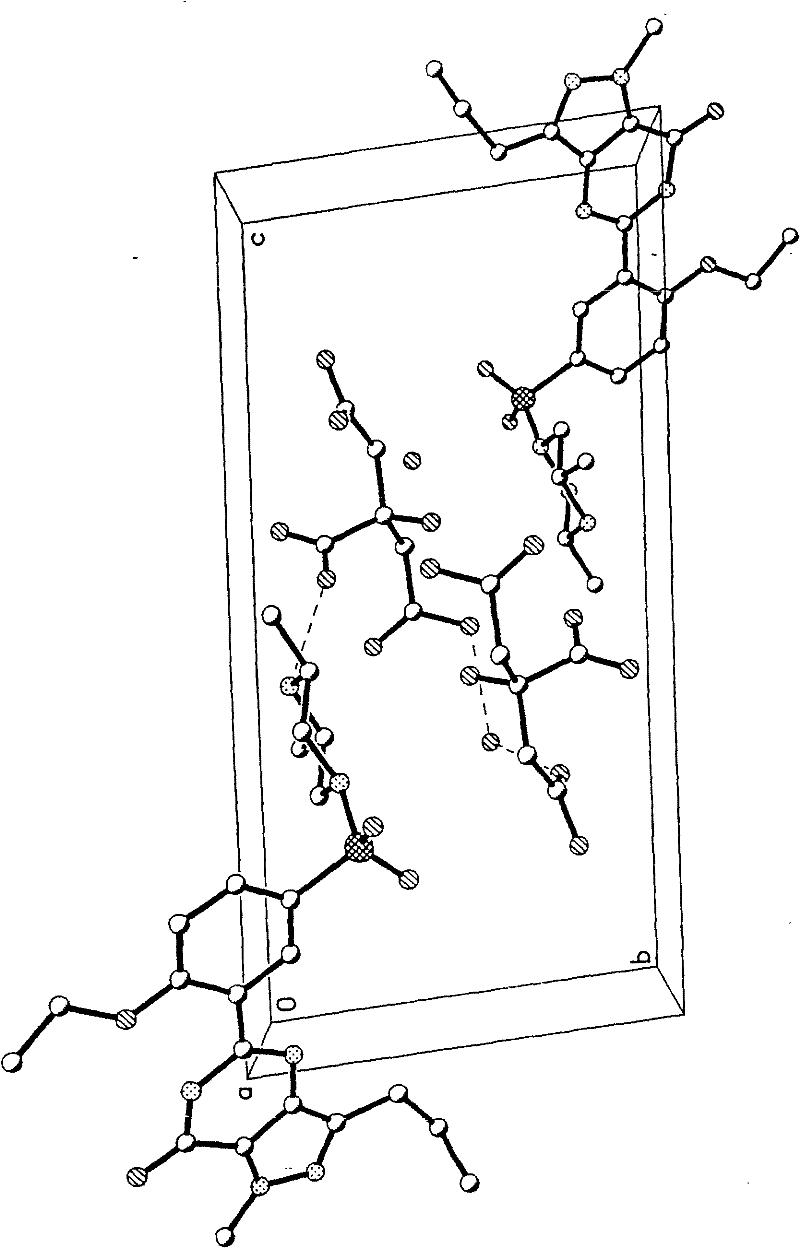
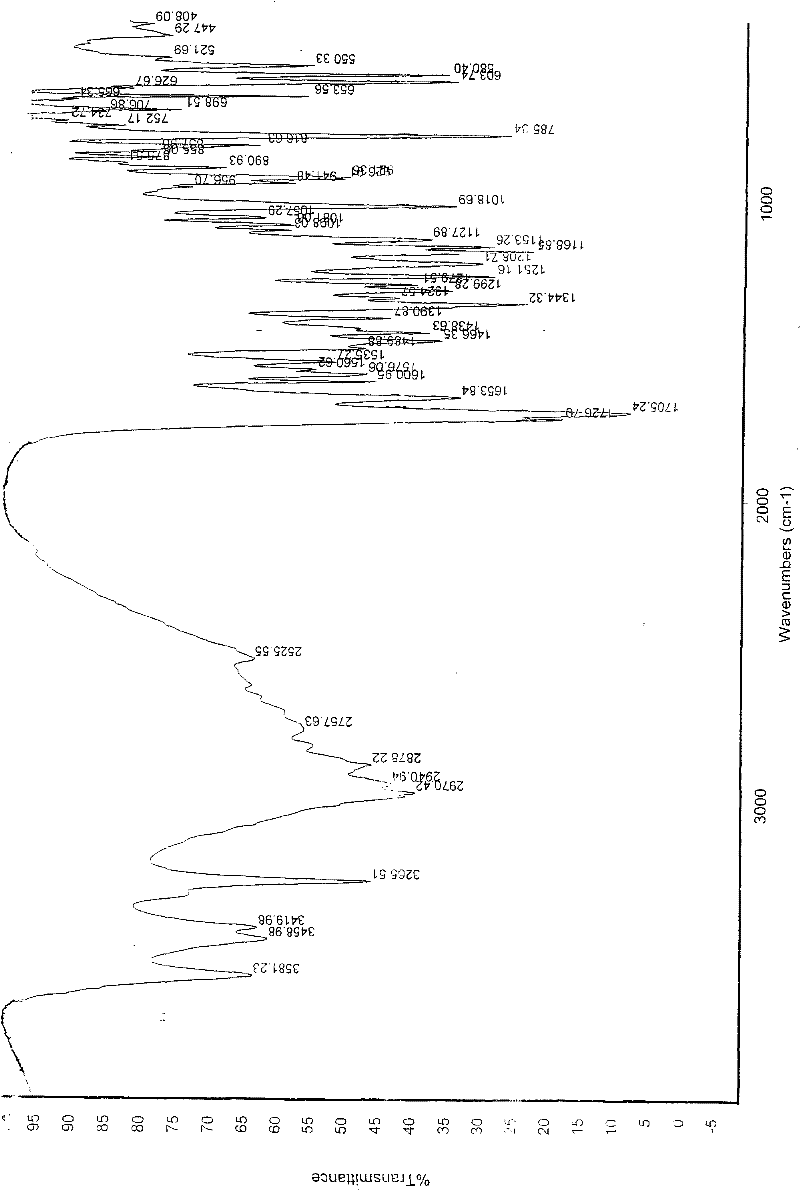
[0094] In a 100ml reaction flask, add 3 grams of aldenafil citrate, 85ml of distilled water / absolute ethanol (50:50, volume ratio), start stirring, heat and heat up to reflux temperature, filter while hot after 15 minutes, seal the container Put the filtrate in a filter bottle, place it indoors, cool down naturally, drop to 30°C-35°C, keep it at a temperature for 58 hours, precipitate out crystals, filter, place indoors for 1 hour, and dry to obtain citrate aldenafil crystalline form D 1.8g, its melting point is m.p 201.9-203.1°C, the purification rate is 60%, and the content measured by HPLC area normalization method is 99.92%. See figure 1 , figure 2 , image 3 and Image 6 , are the three-dimensional structure projection diagram, unit cell projection diagram, infrared spectrum and X-ray powder diffraction diagram of aldenafil citrate crystalline form D, showing the characteristics of citrate aldenafil crystalline form D.
Embodiment 2
[0096] In a 200ml reaction flask, add 5 grams of aldenafil citrate, 160ml of distilled water / absolute ethanol (40:60, volume ratio), start stirring, heat up to reflux temperature, filter while hot after 15 minutes, seal Put the filter bottle of the filtrate, place it indoors, cool down naturally, drop it to 30°C-35°C, keep it at a temperature for 65 hours, separate out crystals, filter, put it indoors for 1 hour, and dry it to obtain citrate adenafil form D 2.8g, the melting point is m.p 201.9-203.1°C, the purification rate is 56%, and the content measured by the HPLC area normalization method is 99.92%. After testing, it shows the characteristics of idenafil citrate form D.
Embodiment 3
[0098] Granules containing edenafil citrate form D
[0099] Prescription: 50 grams of edinafil citrate form D, 650 grams of lactose, 100 grams of crospovidin, 90 grams of PEG-400000, 135 grams of hydroxypropyl methylcellulose, appropriate amount of distilled water, made into 1000 bags.
[0100] Process: PEG-4000 and citrate adenafil crystal form D are crushed together, passed through an 80-mesh sieve, mixed with other materials to make soft materials with distilled water, granulated, dried at low temperature and then packed into granules.
PUM
| Property | Measurement | Unit |
|---|---|---|
| density | aaaaa | aaaaa |
| melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More