Preparation method of agomelatine I crystal form
A technology of crystal form and crystal plane spacing, which is applied in the field of medicine, can solve the problems of poor reproducibility and unsuitability for industrial production, and achieve the effect of simple operation and good reproducibility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0037] Example 1: Form I of N-(2-(7-methoxy-1-naphthyl)ethyl)-acetamide
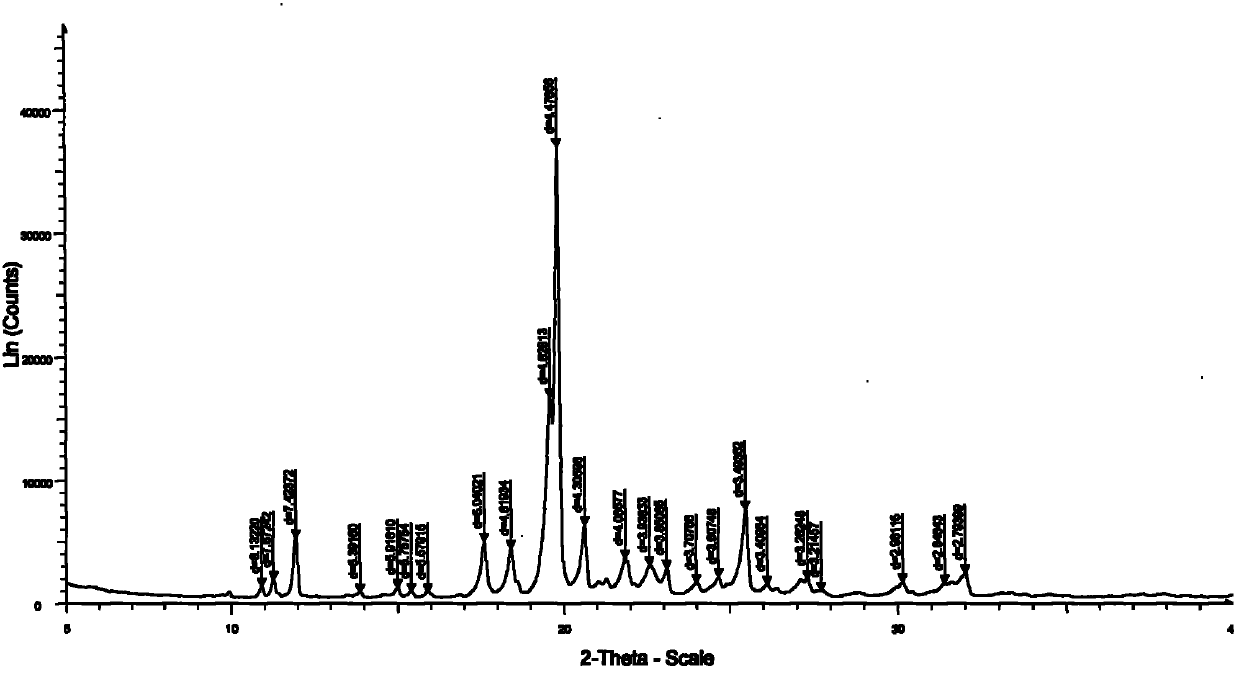
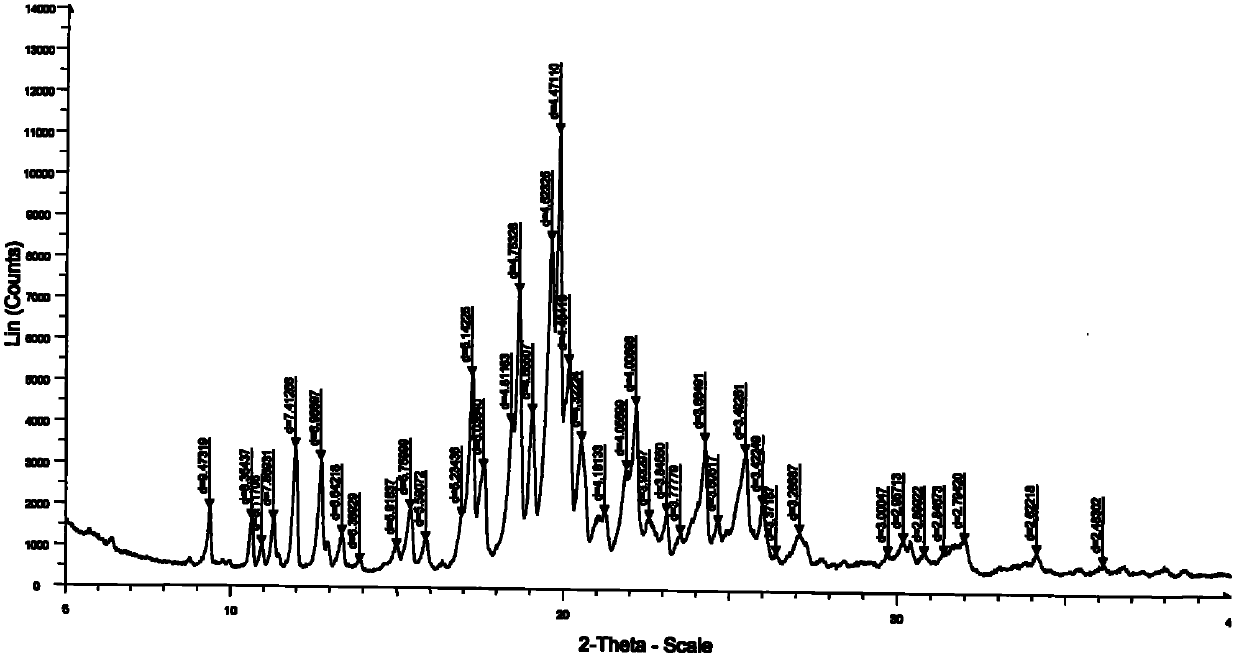
[0038]2 grams of agomelatine sample was added to the flask, followed by a mixed solvent of 2ml N,N-dimethylformamide (DMF) and 3ml water, heated to dissolve, filtered to remove insoluble matter, and the filtrate was cooled to 0 in an ice bath. °C, a solid precipitated out. Suction filtration after 0.5 hours, the filter cake was washed twice with water, dried by suction, and dried in vacuum at 30°C for 12 hours to obtain 1.8 g of agomelatine I crystal form sample with a yield of 90% and a purity of 99.2%. Its DSC spectrum is shown in figure 1 , X-ray powder diffraction pattern see figure 2 .
Embodiment 2
[0039] Example 2: Form I of N-(2-(7-methoxy-1-naphthyl)ethyl)-acetamide
[0040] Add 2 grams of agomelatine sample into the flask, then add 3ml of N,N-dimethylacetamide and 2ml of water to form a mixed solvent, heat to dissolve, filter out insoluble matter, and slowly cool the filtrate to 10 °C, a solid precipitated out. Suction filtration after 3 hours, the filter cake was washed twice with water, dried by suction, and dried under vacuum at 50°C for 12 hours to obtain 1.85 g of agomelatine I crystal form sample with a yield of 92.5% and a purity of 99.7%. Its DSC spectrum is shown in figure 1 , X-ray powder diffraction pattern see figure 2 .
Embodiment 3
[0041] Example 3: Form I of N-(2-(7-methoxy-1-naphthyl)ethyl)-acetamide
[0042] Add 2 grams of agomelatine sample into the flask, then add 3mL of N,N-dimethylformamide and 3mL of water as a mixed solvent, heat to dissolve, then filter with suction to remove insoluble matter, and cool the filtrate in an ice bath At 5°C, a solid precipitated out. Suction filtration after 2 hours, the filter cake was washed twice with water, dried by suction, and dried under vacuum at 40°C for 12 hours to obtain 1.78 g of agomelatine I crystal form sample with a yield of 89% and a purity of 99.5%. Its DSC spectrum is shown in figure 1 , X-ray powder diffraction pattern see figure 2 .
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More