Preparation method of olanzapine
A technology of olanzapine and reaction, applied in the field of preparation of olanzapine, can solve the problems of low yield, dangerous operation, environmental pollution, etc., and achieve the effects of high yield, less environmental pollution, and reduction of three wastes discharge
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030] Example 1, the preparation of 2-(o-nitroanilino)-3-cyano-5-methylthiophene, namely compound III.
[0031] In a 1000ml four-necked flask, add 200ml of DMF, 110g of o-fluoronitrobenzene as compound I, 45g of anhydrous potassium fluoride and 90g of 2-amino-3-cyano-5-methylthiophene as compound II. , heat up to 70~75°C, react for 5~6.0h until the reaction is complete, cool down to 20~30°C, filter, wash the filter cake with a small amount of DMF, combine the filtrate, recover DMF under reduced pressure until dry, add 200ml of absolute ethanol , refluxed for 0.5h, lowered the temperature to 0~5°C, filtered with suction, drained, dug out the solid, and dried under reduced pressure at 50~60°C to obtain 15.5g of a yellow solid, with a molar yield of 0.92 based on compound II, HPLC98.9% .
Embodiment 2
[0032] Example 2, the preparation of 2-methyl-4-amino-10H-thieno[2,3-b][1,5]benzodiazepine sulfate, compound IV.
[0033] In a 2000ml flask, add 80.0g of compound III, 800ml of ethanol with a weight percentage of 95% and 40g of zinc powder, raise the temperature to 50~55°C, keep it warm for 3~4h, after the reaction is complete, cool it down and add 20% by weight of H 2 SO 4 377.0g, then keep warm at 60~65°C for 2~4.0h, follow the completion of the reaction by HPLC, cool slightly, recover ethanol under reduced pressure, cool to 5~10°C to crystallize for 2.0h, filter, and dry the pale yellow solid that is compound IV 90.8g, HPLC 99.5%, molar yield based on compound (III) 0.90.
Embodiment 3
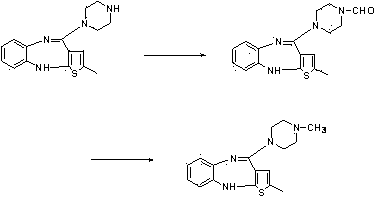
[0034] Embodiment 3, the preparation of olanzapine, namely compound VI.
[0035] Into a 1000 ml four-necked flask, 80 g of compound IV, 500 ml of N-methylpyrrolidone and 196 g of N-methylpiperazine were added. Raise the temperature to 100~120°C, react for 8~10.0h, the reaction is complete, cool slightly, recover N-methylpyrrolidone under reduced pressure, then cool to 50°C, add 400ml of purified water, cool to 20~25°C, filter, wash with appropriate amount of water, After drying, 72.0 g was obtained. Put 82.0 g of the above crude product into 400 ml of dichloromethane, add 2.0 g of activated carbon, reflux to dissolve, filter while hot, recover most of the dichloromethane under reduced pressure, cool and crystallize, and dry to obtain pure compound VI 66.0 g of light yellow solid, HPLC 99.8%, molar yield based on compound IV is 0.857.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More