New synthesis method of N-acyl-N-methyltaurine
A technology of methyl taurine and sodium acyl taurine, which is applied in the fields of sulfonic acid preparation and organic chemistry, and can solve problems such as harsh process conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
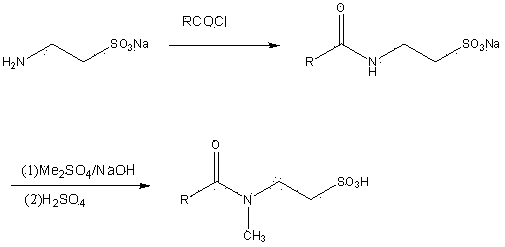
[0017] In a 500 ml three-necked flask, add 150 g (0.25 moles) of 33% sodium taurate aqueous solution, then add 80 g of acetone and stir evenly, cool to below 10°C, add 55 g (0.2 moles) of palmitoyl chloride slowly and uniformly ) At the same time, drop in 50% sodium hydroxide aqueous solution, carefully control the pH of the reaction solution between 9 and 10, and finish the drop in about 1 hour. Continue the reaction at 20-30°C for 2 hours, and the pH should still be 9-10 at the end. The obtained white paste reactant was placed in the refrigerator overnight, filtered to dryness, treated with acetone and dried to obtain 62 grams of white powdery sodium N-palmitoyl taurate (yield 80%).
Embodiment 2
[0019] In a 500 ml three-necked flask, put 39 g (0.1 mole) of sodium N-palmitoyl taurate and 150 ml of water, then add 50 ml of 30% sodium hydroxide, cool to below 10°C, and slowly drop Add 15 g (0.12 mol) of dimethyl sulfate, continue the reaction for 1 hour after the dripping, and then acidify with sulfuric acid to pH 2-3. The precipitated solid is filtered, washed with water and dried to obtain a white powdery N-palmitoyl-N- 25 grams of methyl taurine (90% yield).
Embodiment 3
[0021] In a 500 ml three-necked flask, add 150 g (0.25 mol) of sodium taurate solution with a concentration of 33%, cool to below 10°C, slowly and uniformly add 55 g (0.2 mol) of palmitoyl chloride, and at the same time, drop in 50% aqueous sodium hydroxide solution, carefully control the pH of the reaction solution between 9-10, about 1 hour after dripping, continue to react at 20-30℃ for 2 hours, and the pH should still be 9-10 at the end. Then add 50 ml of 30% sodium hydroxide, cool to below 10°C, slowly add 30 g (0.24 mol) of dimethyl sulfate dropwise, continue the reaction for 1 hour after the dropwise addition, and then acidify with sulfuric acid to pH 2 ~ 3, the precipitated solid was filtered, washed with water and dried to obtain 49 grams of white powdery N-palmitoyl-N-methyl taurine (yield 70%).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More