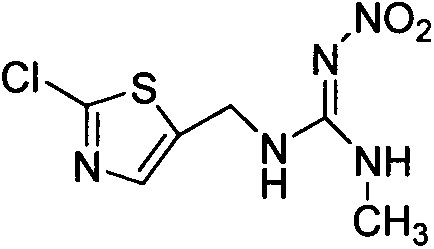
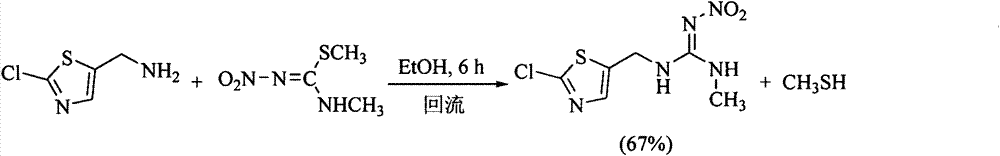
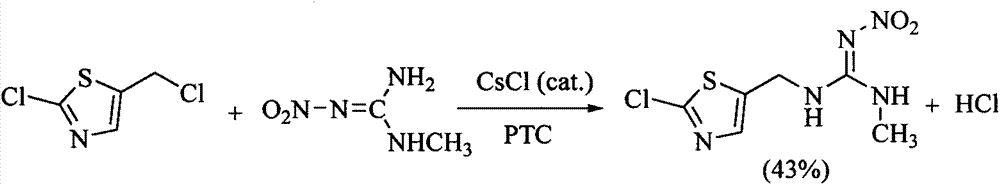
Clothianidin synthesis process
A synthetic process, clothianidin technology, applied in the field of industrial production of pesticides and insecticides, can solve problems such as unfavorable industrial production
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0029] In a 250ml four-necked flask, add DMF (150ml), 1-methyl-5-n-propyl-2-nitroimino-hexahydro-1,3,5-hexahydro-triazine (15.97g , 79mmol), potassium carbonate (27.08g, 196mmol), control the temperature at 20~25°C, add dropwise 2-chloro-5-chloromethylthiazole (13.35g, 79mmol) dissolved in DMF within 30min, after the dropwise addition, add The temperature rises slowly to 40-50°C, traced by high performance liquid chromatography - the content of methyl-5-n-propyl-2-nitroimino-hexahydro-1,3,5-hexahydro-triazine is less than 1.0% , stop the reaction. After filtering while hot, add saturated brine and dichloromethane (volume ratio 4.1:1) to the filtrate at 20-25°C, stir, extract and separate, filter the organic phase, cool the filtrate to crystallize below 0°C, and obtain 1-( 2-chlorothiazole-5-methyl)-2-nitroimino-3-methyl-5-n-propyl-hexahydro-1,3,5-triazine 21.86g, yield 91.0%, purity 99.3% (HPLC), and then generate clothianidin under the action of dilute hydrochloric acid, wi...
Embodiment 2
[0031] In a 250ml four-necked flask, add DMF (150ml), 1-methyl-5-n-propyl-2-nitroimino-hexahydro-1,3,5-hexahydro-triazine (15.97g , 79mmol), potassium carbonate (27.08g, 196mmol), control the temperature at 20~25°C, add dropwise 2-chloro-5-chloromethylthiazole (13.35g, 79mmol) dissolved in DMF within 30min, after the dropwise addition, add The temperature rises slowly to 40-50°C, traced by high performance liquid chromatography - the content of methyl-5-n-propyl-2-nitroimino-hexahydro-1,3,5-hexahydro-triazine is less than 1.0% , stop the reaction. After filtering while hot, add saturated brine and ethyl acetate (volume ratio 4.1:1) to the filtrate at 20-25°C, stir, extract and separate, filter the organic phase, and cool the filtrate to crystallize below 0°C, and obtain 1-( 2-chlorothiazole-5-methyl)-2-nitroimino-3-methyl-5-n-propyl-hexahydro-1,3,5-triazine 24.01g, yield 95.5%, purity 99.7% (HPLC), and then generate Clothianidin under the action of dilute hydrochloric acid, ...
Embodiment 3
[0033]In a 250ml four-necked flask, add DMF (150ml), 1-methyl-5-n-propyl-2-nitroimino-hexahydro-1,3,5-hexahydro-triazine (15.97g , 79mmol), potassium carbonate (27.08g, 196mmol), control the temperature at 20~25°C, add dropwise 2-chloro-5-chloromethylthiazole (13.35g, 79mmol) dissolved in DMF within 30min, after the dropwise addition, add The temperature rises slowly to 40-50°C, traced by high performance liquid chromatography - the content of methyl-5-n-propyl-2-nitroimino-hexahydro-1,3,5-hexahydro-triazine is less than 1.0% , stop the reaction. After filtering while hot, add saturated brine and petroleum ether (volume ratio 4.1:1) to the filtrate at 20-25°C, stir for extraction and separation, filter the organic phase, cool the filtrate to crystallize below 0°C, and suction filter to obtain 1-(2 -Chlorothiazole-5-methyl)-2-nitroimino-3-methyl-5-n-propyl-hexahydro-1,3,5-triazine yield 23.15g, 92.14%, purity 99.3 % (HPLC), and then generate clothianidin under the effect of d...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More