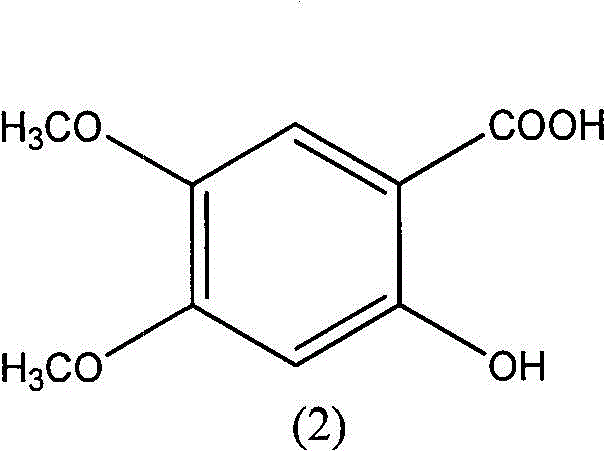
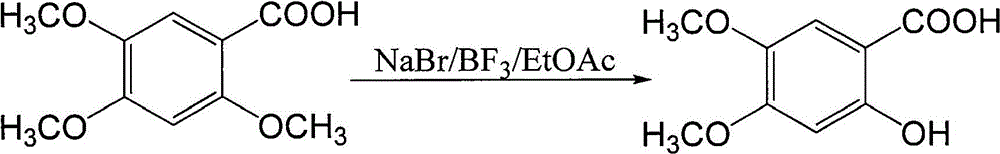Method for preparing 2-hydroxy-4,5-dimethoxybenzoic acid
A technology of trimethoxybenzoic acid and dimethylacetamide, applied in the field of preparing intermediate 2-hydroxyl-4, which can solve the problem of strong volatility that is not conducive to labor protection, metal Lewis acid environmental problems, and the price of boron trifluoride ether More expensive and other problems, to achieve the effect of less impurities, short reaction time, suitable for industrial production
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
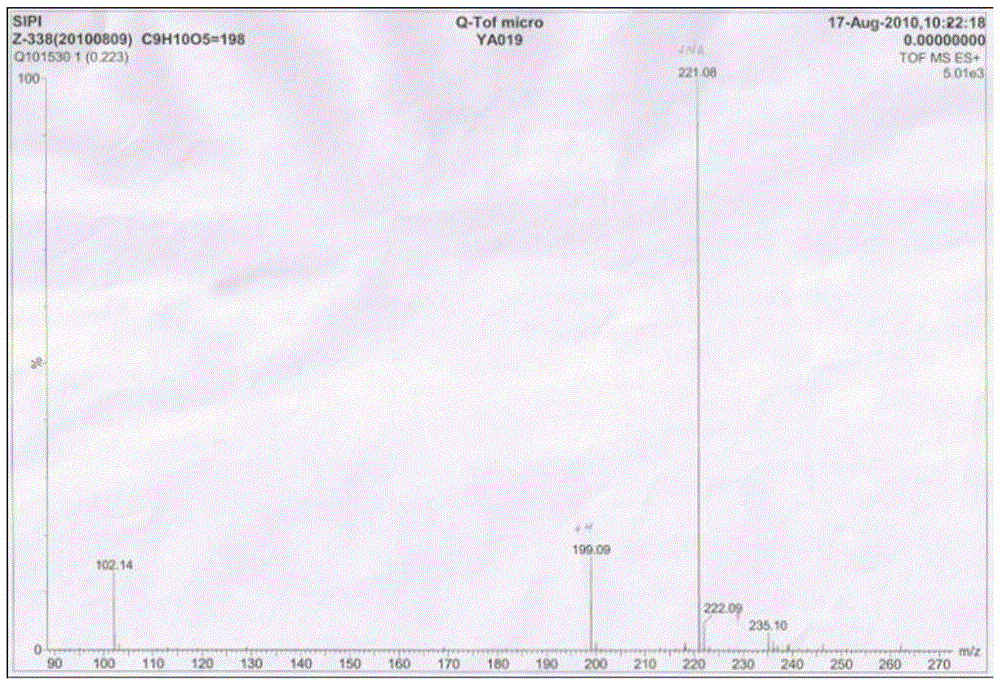
[0020] N 2 Under air flow, 2.0 g of 2,4,5-trimethoxybenzoic acid and 5.45 g of piperazine were suspended in 10 ml of N,N-dimethylacetamide, heated and stirred at 150° C. for 2 hours to stop the reaction. 70°C water pump rotary steaming to constant weight, and then oil pump rotary steaming to constant weight. Adjust the pH, stir overnight at room temperature, filter with suction, and dry under reduced pressure to obtain 1.81 g of the product with a yield of 95.3%. m + 199, M+Na + 221.
Embodiment 2
[0022] N 2 Under air flow, 2.0 g of 2,4,5-trimethoxybenzoic acid and 5.45 g of piperazine were suspended in 10 ml of N,N-dimethylacetamide, heated and stirred at 140° C. for 1 hour to stop the reaction. 70°C water pump rotary steaming to constant weight, and then oil pump rotary steaming to constant weight. Adjust the pH, stir overnight at room temperature, filter with suction, and dry under reduced pressure to obtain 1.4 g of the product with a yield of 75%.
Embodiment 3
[0024] N 2 Under air flow, 2.0 g of 2,4,5-trimethoxybenzoic acid and 5.5 g of morpholine were suspended in N,N-dimethylacetamide, heated and stirred at 150° C. for 3 hours to stop the reaction. 70°C water pump rotary steaming to constant weight, and then oil pump rotary steaming to constant weight. Adjust the pH, stir overnight at room temperature, filter with suction, and dry under reduced pressure to obtain 1.5 g of the product with a yield of 80%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More