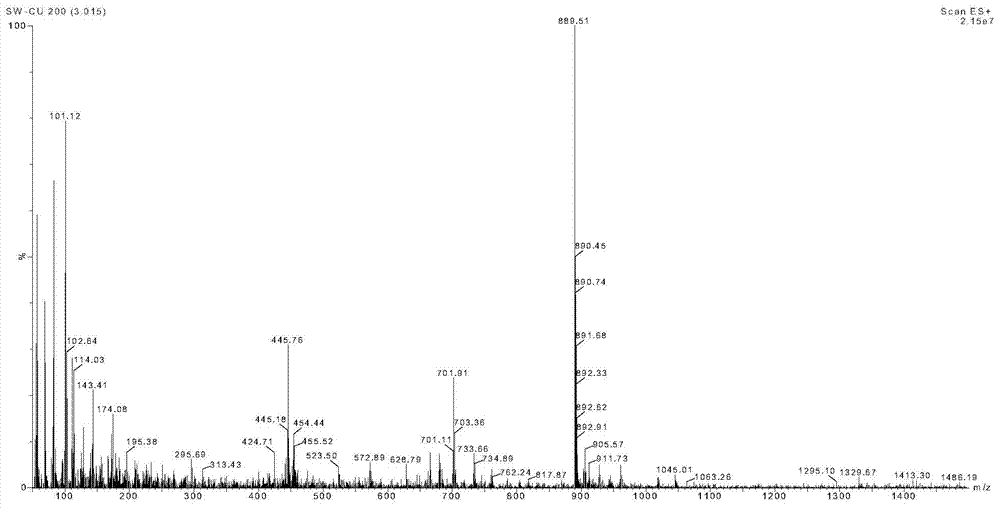
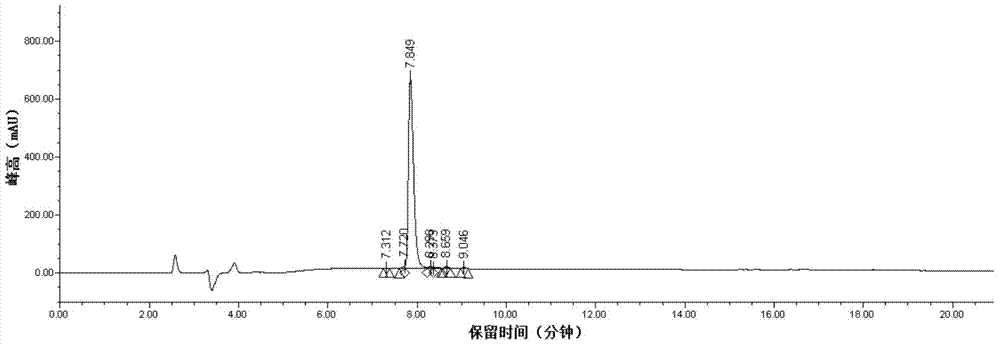
Liquid-phase segmented synthesis method of argireline
A segmented synthesis and hexapeptide technology, which is applied to the preparation method of peptides, chemical instruments and methods, peptides, etc., can solve the problems of excessive reaction reagents, high prices, and many types of solvents.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0038] 1. Synthesis of AcOSu
[0039] According to the molar ratio of N-hydroxysuccinimide to diisopropylethylamine and acetyl chloride is 1:1.2:1.1, put 11.5g of N-hydroxysuccinimide in a 250mL three-necked bottle, add 100mL di Chloromethane, mix well, add 20.7mL diisopropylethylamine under stirring condition, cool down to 0~5℃ in ice bath, when the solution becomes clear, add 7.9mL acetyl chloride dropwise, keep the solution temperature below 20°C during the dropwise addition ℃, remove the ice bath after the dropwise addition, naturally rise to room temperature and stir for 3 hours, wash the reaction solution with water 3 times, 50mL each time, dry the organic phase over anhydrous sodium sulfate, filter, and concentrate under reduced pressure to obtain 15.3g of AcOSu , the yield is 97.4%.
[0040] 2. Synthesis of Ac-Glu(OtBu)-OH
[0041] According to the molar ratio of AcOSu to γ-tert-butyl glutamic acid and sodium bicarbonate as 1:1.1:2, and the volume ratio of distilled ...
Embodiment 2
[0066] Synthesis of Ac-Glu(OtBu)-OH step 2, Synthesis of Ac-Glu(OtBu)-Glu(OtBu)-OH step 4, Synthesis of Ac-Glu(OtBu)-Glu(OtBu)-Met-OH step in this example 6. In step 9 of the synthesis of Fmoc-Gln(Trt)-Arg-OH, the molar weight of sodium bicarbonate used is 1.5 times that of sodium bicarbonate in Example 1. Other steps are identical with embodiment 1, obtain 6.4g hexapeptide, yield is 90.1%, HPLC detection shows that purity is 90%.
Embodiment 3
[0068] Synthesis of Ac-Glu(OtBu)-OH step 2, Synthesis of Ac-Glu(OtBu)-Glu(OtBu)-OH step 4, Synthesis of Ac-Glu(OtBu)-Glu(OtBu)-Met-OH step in this example 6. In step 9 of synthesizing Fmoc-Gln(Trt)-Arg-OH, the molar weight of sodium bicarbonate used is 3 / 4 of the molar weight of sodium bicarbonate in Example 1. The other steps were the same as in Example 1 to obtain 6.47g hexapeptide with a yield of 91%, and HPLC detection showed that the purity was greater than 90%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More