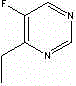
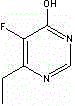
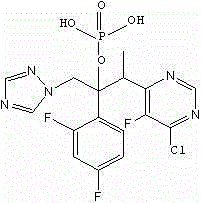
HPLC method for separating and analyzing voriconazole prodrug related substances
A technology for voriconazole and related substances, which is applied in the field of high performance liquid phase analysis and can solve problems such as difficulty in effective separation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0056] Experimental equipment and conditions
[0057] High performance liquid chromatography: Thermo high performance liquid chromatography Ultimate3000;
[0058] Chromatographic column: C18150×4.6mm5μm;
[0059] Mobile phase: acetonitrile: buffer (take 0.606g of sodium heptanesulfonate and 6.80g of potassium dihydrogen phosphate into 1000ml of water, stir to dissolve, adjust the pH to 3.00 with phosphoric acid) = 20:80;
[0060] Flow rate 1.0ml / min;
[0061] Detection wavelength: 256nm;
[0062] Column temperature 20°C;
[0063] The injection volume was 10 μl.
[0064]Get voriconazole phosphate or its medicinal salt and the above-mentioned impurities, dissolve and dilute with a suitable medium to make the test solution and the impurity solution, get each 10 μl of the test solution and the impurity solution, inject into the high performance liquid chromatograph, the results are as follows:
[0065] sample Retention time (min) Separation Impurity I 4.83...
Embodiment 2
[0068] Experimental equipment and conditions
[0069] High performance liquid chromatography: Thermo high performance liquid chromatography Ultimate3000;
[0070] Chromatographic column: C18150×4.6mm5μm;
[0071] Mobile phase: Phase A: buffer solution (take 0.606g of sodium heptanesulfonate and 6.80g of potassium dihydrogen phosphate into 1000ml of water, stir to dissolve, adjust the pH to 3.00 with phosphoric acid); phase B: acetonitrile; the gradient conditions are as follows:
[0072] time (min) Phase A (%) Phase B (%) 0 80 20 15 80 20 45 50 50 50 82 18 60 82 18
[0073] Flow rate 1.0ml / min;
[0074] Detection wavelength: 256nm;
[0075] Column temperature 25°C;
[0076] The injection volume was 10 μl.
[0077] Get voriconazole phosphate or its medicinal salt and the above-mentioned impurities, dissolve and dilute with a suitable medium to make the test solution and the impurity solution, get each 10 μl of the test solut...
Embodiment 3
[0081] Experimental equipment and conditions
[0082] High performance liquid chromatography: Thermo high performance liquid chromatography Ultimate3000;
[0083] Chromatographic column: phenyl column 250×4.6mm5μm;
[0084] Mobile phase: Phase A: buffer solution (take 0.606g of sodium heptanesulfonate and 6.80g of potassium dihydrogen phosphate into 1000ml of water, stir to dissolve, adjust the pH to 3.00 with phosphoric acid); phase B: acetonitrile; the gradient conditions are as follows:
[0085] time (min) Phase A (%) Phase B (%) 0 82 18 15 82 18 45 50 50 50 82 18 60 82 18
[0086] Flow rate 1.0ml / min;
[0087] Detection wavelength: 256nm;
[0088] Column temperature 25°C;
[0089] The injection volume was 10 μl.
[0090] Get voriconazole phosphate or its medicinal salt and the above-mentioned impurities, dissolve and dilute with a suitable medium to make the test solution and the impurity solution, get each 10 μl of the t...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Wavelength | aaaaa | aaaaa |
| Wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More