A kind of preparation method of everolimus
A technology of everolimus and intermediates, applied in the field of preparation of everolimus, which can solve the problems of insufficient reaction, low total product yield, unstable properties, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
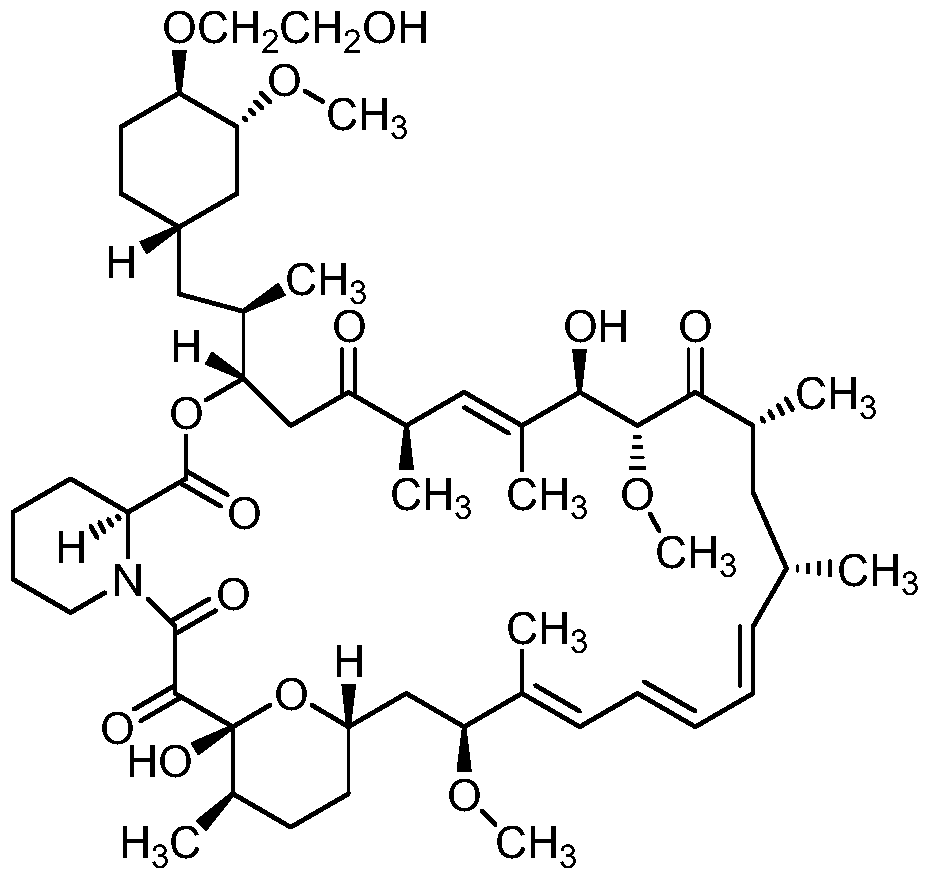
[0028] Embodiment 1: Preparation of Everolimus Intermediate 1:
[0029] Add 9g of rapamycin, 4.5g of 2,6-lutidine, and 100ml of tetrahydrofuran into a 250ml three-neck flask, cool down in an ice-salt water bath, stir to dissolve, and start to drop 7ml of (CH 3 ) 3 SiCl, heat preservation reaction, after 3 hours of reaction, TLC detects the reaction process, if there is no rapamycin spot, then the double protection reaction is completed, then add 5ml HCl dropwise, after 5 hours of reaction, TLC detection, if there is no double protection rapamycin spot, then The hydrolysis reaction is complete. The reaction solution was extracted with 200ml of ethyl acetate, and the ethyl acetate layer was extracted with 50ml of water, NaHCO 3 Wash with 50ml of saturated solution, then wash with 50ml of salt water, extract the aqueous layer twice with ethyl acetate, combine the organic layers, and anhydrous Na 2 SO 4 Drying; Ethyl acetate was evaporated under reduced pressure to obtain 9.6 ...
Embodiment 2
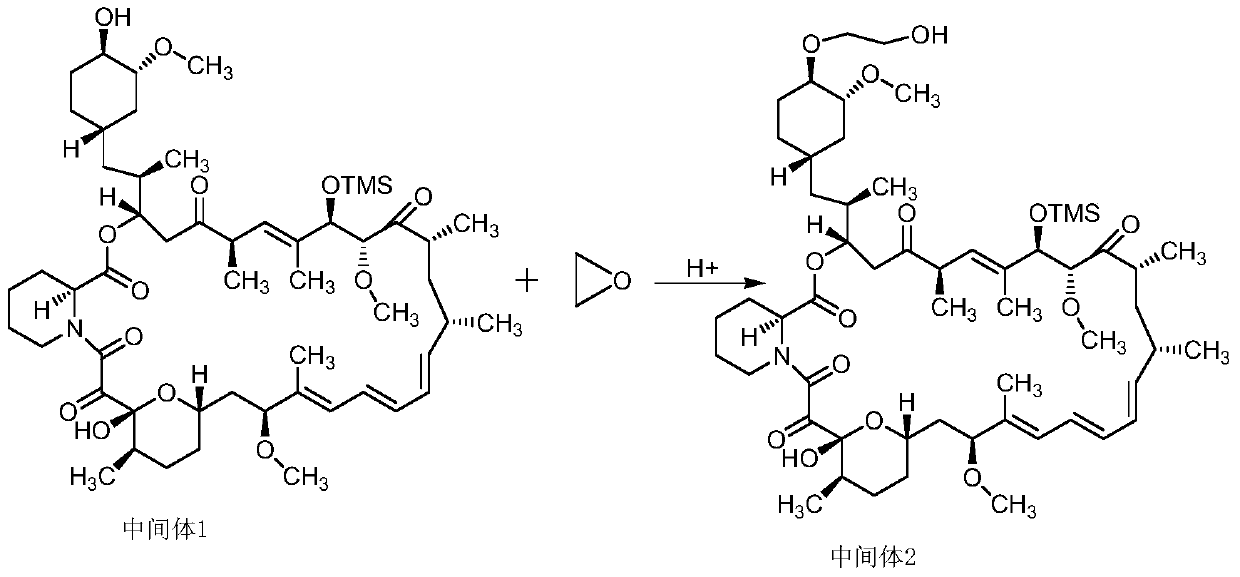
[0030] Embodiment 2: Preparation of Everolimus Intermediate 2
[0031] At 0-10°C, add 500ml of toluene to a 1L three-necked round-bottomed flask, then add 4.4g of ethylene oxide, after cooling down, add 9.6g of rapamycin intermediate 1 into the reaction solution, and add a small amount of hydrochloric acid at the same time, Continue to react for 6 hours, TLC monitors until the reaction is complete, water 250ml, NaHCO 3 Wash with 250ml of saturated solution, then wash with 250ml of salt water, extract the aqueous layer twice with toluene, combine the organic layers, and anhydrous Na 2 SO 4 Drying; toluene was evaporated under reduced pressure to obtain 6.8 g of pure everolimus intermediate 2.
Embodiment 3
[0032] Embodiment 3: Preparation of everolimus crude product
[0033] Dissolve 6.8g of the coupled product in 500ml of methanol in a 1L single-necked bottle, cool down in an ice-water bath, add 10ml of HCl dropwise after half an hour, react for 4 hours after the dropwise addition, and track and detect with TLC. After the reaction is completed, evaporate under reduced pressure Dry, then dissolve the residue with 500ml ethyl acetate, wash with saturated brine, extract the aqueous layer 3 times with ethyl acetate, combine the organic layers, dry over anhydrous sodium sulfate, evaporate to dryness under reduced pressure to obtain a yellow oil, and separate through the column to obtain the crude product 5.6g.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More