Metal Cu (I) complex, preparation method and application thereof
A complex and metal technology, applied in organic compound/hydride/coordination complex catalysts, copper organic compounds, chemical instruments and methods, etc. The effect of low preparation cost and mild reaction conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
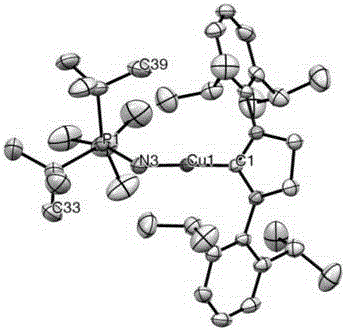
[0044] Embodiment 1: the preparation of metal Cu (I) complex (catalyst) A and B
[0045] 1) Dissolve 0.2mmol (NHC) CuCl in 3ml dry THF in a 20ml dry glass bottle to form a suspension, then put the glass bottle into a glove box refrigerator and freeze at -35°C for 1h;
[0046] 2) In another 20ml dry glass bottle, 42.3mg (0.95eq) [ t Bu 3 PN]Li was dissolved in 3ml of dry THF, and then the glass bottle was placed in a glove box refrigerator and frozen at -35°C for 1h;
[0047] 3) Put 2) the cold [ t Bu 3 PN]Li solution was slowly added dropwise to the cold (NHC) CuCl suspension in 1) under stirring, and continued to stir in the glove box at room temperature for 13h after the dropwise addition;
[0048] 4) After the reaction is complete, dry the solvent (THF) under vacuum to obtain an oily liquid residue, then add 7ml of n-pentane or n-hexane to obtain a suspension, and stir at room temperature for 15min, then pass through neutral diatomaceous earth for a short time Pad filt...
Embodiment 2
[0053] Example 2: Complex B was used to catalyze the hydroboration reaction of phenylacetylene compounds to obtain product 2a with a yield of 89%.
[0054]
Embodiment 3
[0055] Example 3: Complex B was used to catalyze the hydroboration reaction of p-chlorophenylacetylene to obtain product 2b with a yield of 99%.
[0056]
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More