Amorphous telmisartan-pimelic acid eutectic crystal and preparation method and application thereof
A technology of telmisartan and pimelic acid is applied in the field of medicine to achieve the effects of improving bioavailability, low production cost and simple preparation method
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0053] Put 51.4mg (0.1mmol) telmisartan raw material and 15.8mg (0.1mmol) pimelic acid into a mortar, drop a few drops of ethanol at room temperature, and grind for 3 to 4 hours to obtain 67.2mg of amorphous telmisartan Cocrystal of sartan and pimelic acid.
Embodiment 2
[0055] Put 102.8 mg (0.2 mmol) of telmisartan raw material and 31.6 mg (0.2 mmol) of pimelic acid into a mortar, drop a few drops of ethanol at room temperature, and grind for 3 to 4 hours to obtain 134.4 mg of amorphous telmisartan Cocrystal of sartan and pimelic acid.
Embodiment 3
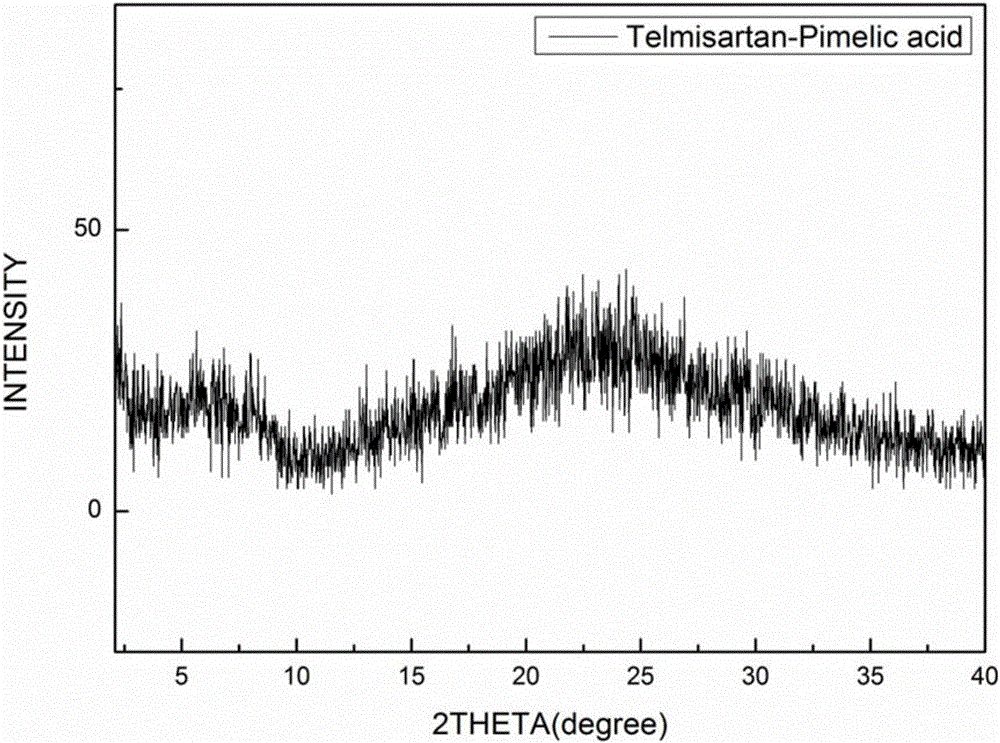
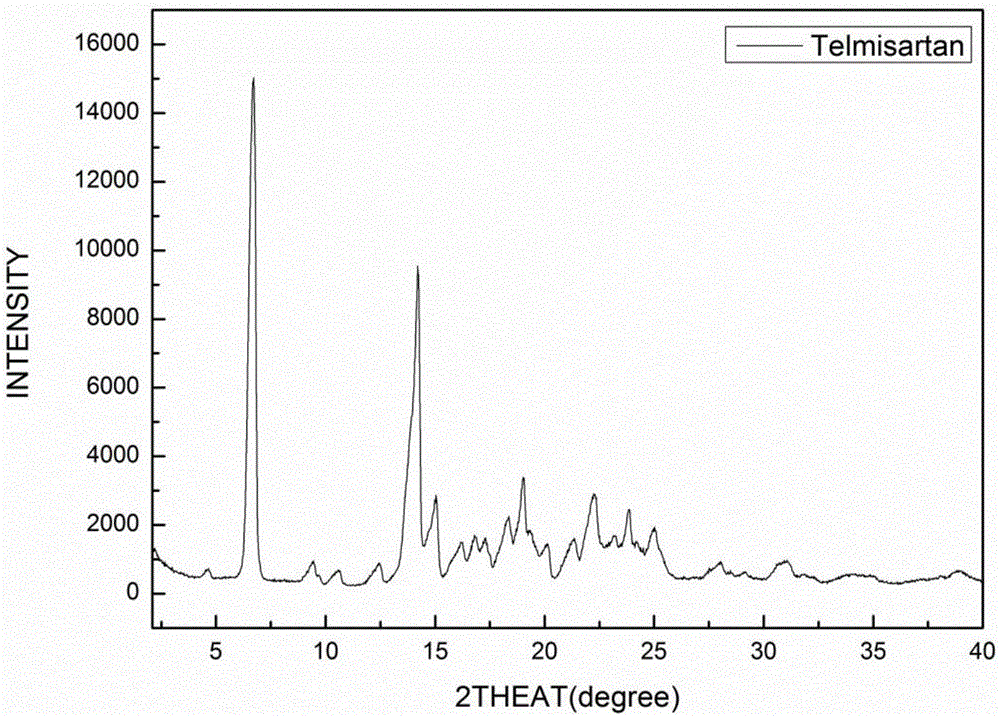
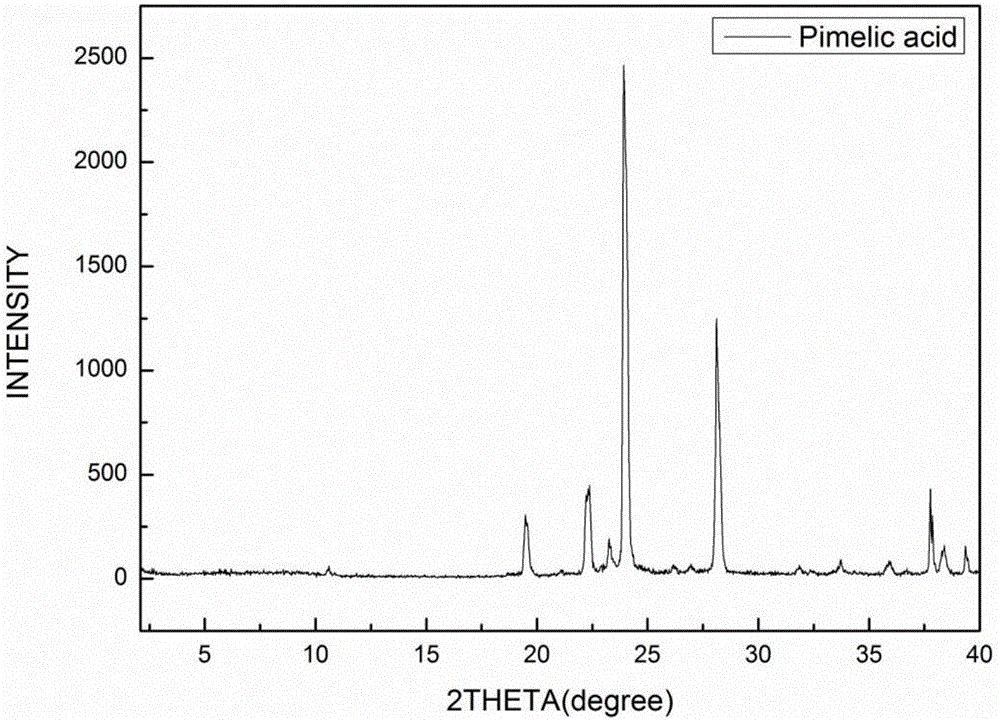
[0057] The eutectic of the amorphous telmisartan prepared in Example 1 and pimelic acid uses Cu-kα radiation, and its X-ray diffraction pattern is as follows: figure 1 shown. Specifically, a Bruker D8 Advance diffractometer was used, and the measurement conditions were as follows: Cu-Kα, 40KV, 40mV light source, step size 0.02°, scanning speed 1° / min, scanning range 2-40°, room temperature. The X-ray diffraction pattern of Telmisartan is as follows figure 2 As shown, the X-ray diffraction pattern of pimelic acid is image 3 shown.
[0058] The eutectic infrared absorption spectrum of the amorphous telmisartan prepared in embodiment 1 and pimelic acid is as follows Figure 4 shown. The wave numbers of its IR characteristic peaks are: 3060.0, 3029.7, 2933.4, 2871.1, 2520.5, 1706.4, 1616.5, 1599.1, 1567.7, 1515.8, 1459.8, 1409.6, 1334.9, 1230.1, 1131.8, 1089.08, 1458.9.6 The carboxyl characteristic peak wave number of telmisartan is 1694.7, while in the infrared spectrum of...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More