Alkoxylated silicified perfume precursor as well as preparation method and application thereof
A technology of alkoxy silicon and deodorant, which is applied in the field of alkoxy siliconized pre-fragrance and its preparation, which can solve the problems of complex preparation process, poor material safety and limited application, and achieve broad development prospects and stable structure
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
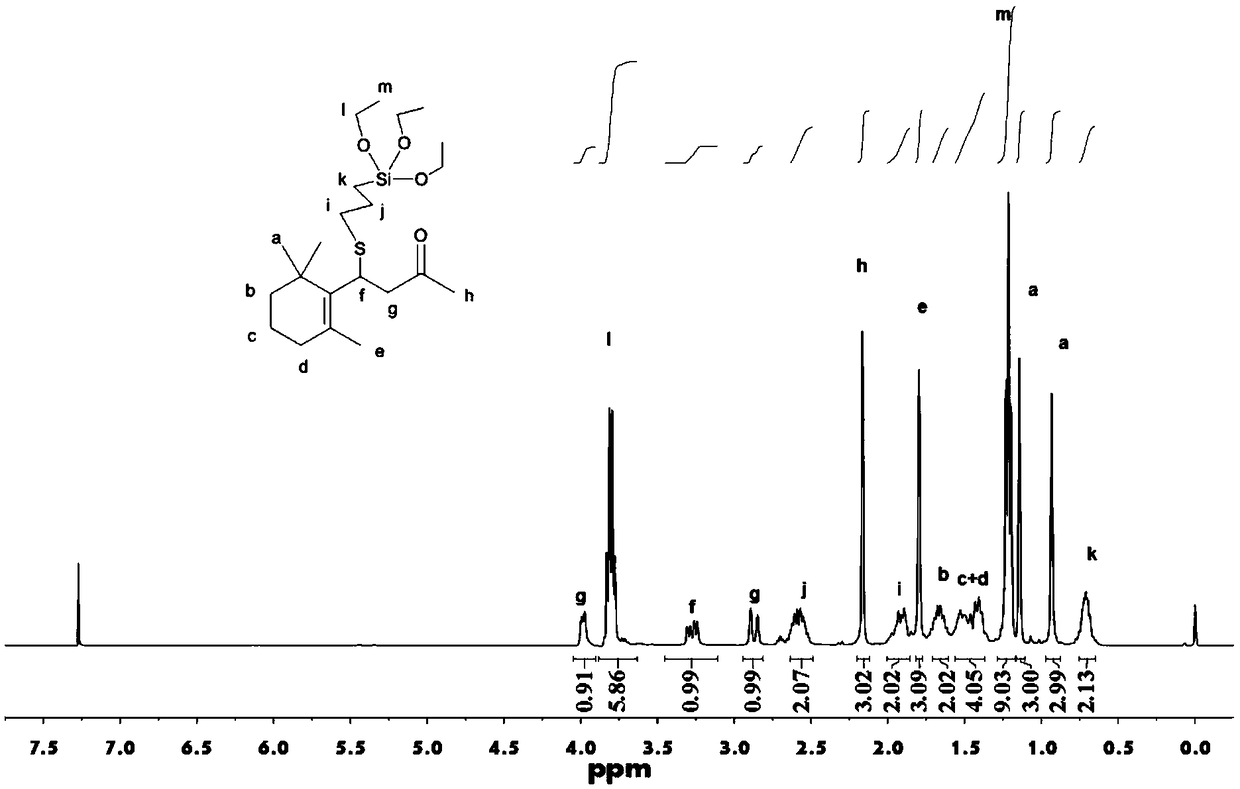
[0032] Mix and react β-ionone, γ-mercaptopropyltriethoxysilane and 1,8-diazabicycloundec-7-ene (DBU) in an organic solvent, and the ratio of the three reactants It is 10g β-ionone and 14.8g γ-mercaptopropyltriethoxysilane (molar ratio 1:1.2) and 250mg DBU. The reaction conditions are anhydrous, oxygen-free, and nitrogen protection, the reaction temperature is 30°C, and the reaction time is 72h. The yellow viscous liquid is dissolved in an organic solvent, saturated sodium bicarbonate and sodium chloride for extraction treatment, acidified with dilute acid, dried with anhydrous magnesium sulfate, and spin-dried to obtain a viscous liquid, which is then distilled under reduced pressure to obtain alkoxy base siliconized pre-fragrance body (yield 82%), its NMR spectrum is as figure 1 , 2 ,Data are as follows:
[0033] 1 H-NMR (CDCl 3 ,400MHz) δ(ppm): 0.70(m, 2H), 0.92(s, 3H), 1.13(s, 3H), 1.19-1.23(m, 9H), 1.42-1.50(m, 4H), 1.66(m , 2H), 1.79(s, 3H), 1.87-1.92(m, 2H), 2.16(s...
Embodiment 2
[0036] Mix and react α-ionone, γ-mercaptopropyltriethoxysilane and 1,8-diazabicycloundec-7-ene (DBU) in an organic solvent, and the ratio of the three reactants It is 10g α-ionone and 14.8g γ-mercaptopropyltriethoxysilane (molar ratio 1:1.2) and 250mg DBU. The reaction conditions are anhydrous, oxygen-free, and nitrogen protection, the reaction temperature is constant at 60° C., and the reaction time is 72 hours. The yellow viscous liquid is dissolved in an organic solvent, saturated sodium bicarbonate and sodium chloride for extraction treatment, acidified with dilute acid, dried with anhydrous magnesium sulfate, and spin-dried to obtain a viscous liquid, which is then distilled under reduced pressure to obtain alkoxy Base siliconized pre-fragrance body (yield 85%), its nuclear magnetic spectrum data are as follows:
[0037] 1 H-NMR (CDCl 3 ,400MHz) δ(ppm): 0.56(m, 2H), 0.93(s, 3H), 1.11(s, 3H), 1.18-1.21(m, 9H), 1.42-1.56(m, 2H), 1.62(m , 2H), 1.66(s, 3H), 1.87-1.93(m, 1...
Embodiment 3
[0040] α-Damascenone, γ-mercaptopropyltriethoxysilane and 1,8-diazabicycloundec-7-ene (DBU) were mixed and reacted in an organic solvent, and the three reactants were formulated The ratio was 4.8g α-ionone to 6.3g γ-mercaptopropyltriethoxysilane (1:1 molar ratio) and 250mg DBU. The reaction conditions are anhydrous, oxygen-free, and nitrogen protection, the reaction temperature is constant at 70° C., and the reaction time is 72 hours. The yellow viscous liquid is dissolved in an organic solvent, saturated sodium bicarbonate and sodium chloride for extraction treatment, acidified with dilute acid, dried with anhydrous magnesium sulfate, and spin-dried to obtain a viscous liquid, which is then distilled under reduced pressure to obtain alkoxy Base siliconized pre-fragrance body (yield 90%), its nuclear magnetic spectrum data are as follows:
[0041] 1 H-NMR (CDCl 3 ,400MHz) δ(ppm): 0.56(m, 2H), 0.93(s, 3H), 1.11(s, 3H), 1.18-1.21(m, 9H), 1.75(s, 3H), 1.48-1.62(m , 4H), 1.93(...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com