Ipratropium bromide aerosol composition without propellant and preparation method thereof
A technology of ipratropium bromide and composition, applied in the field of preparation of the medicine
Pending Publication Date: 2019-10-08
TIANJIN JINYAO GRP
View PDF3 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
[0013] Lysine is a common drug, especially amino acid can be used as a carrier in dry powder inhalation (powder aerosol), which has not been reported in aerosol
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
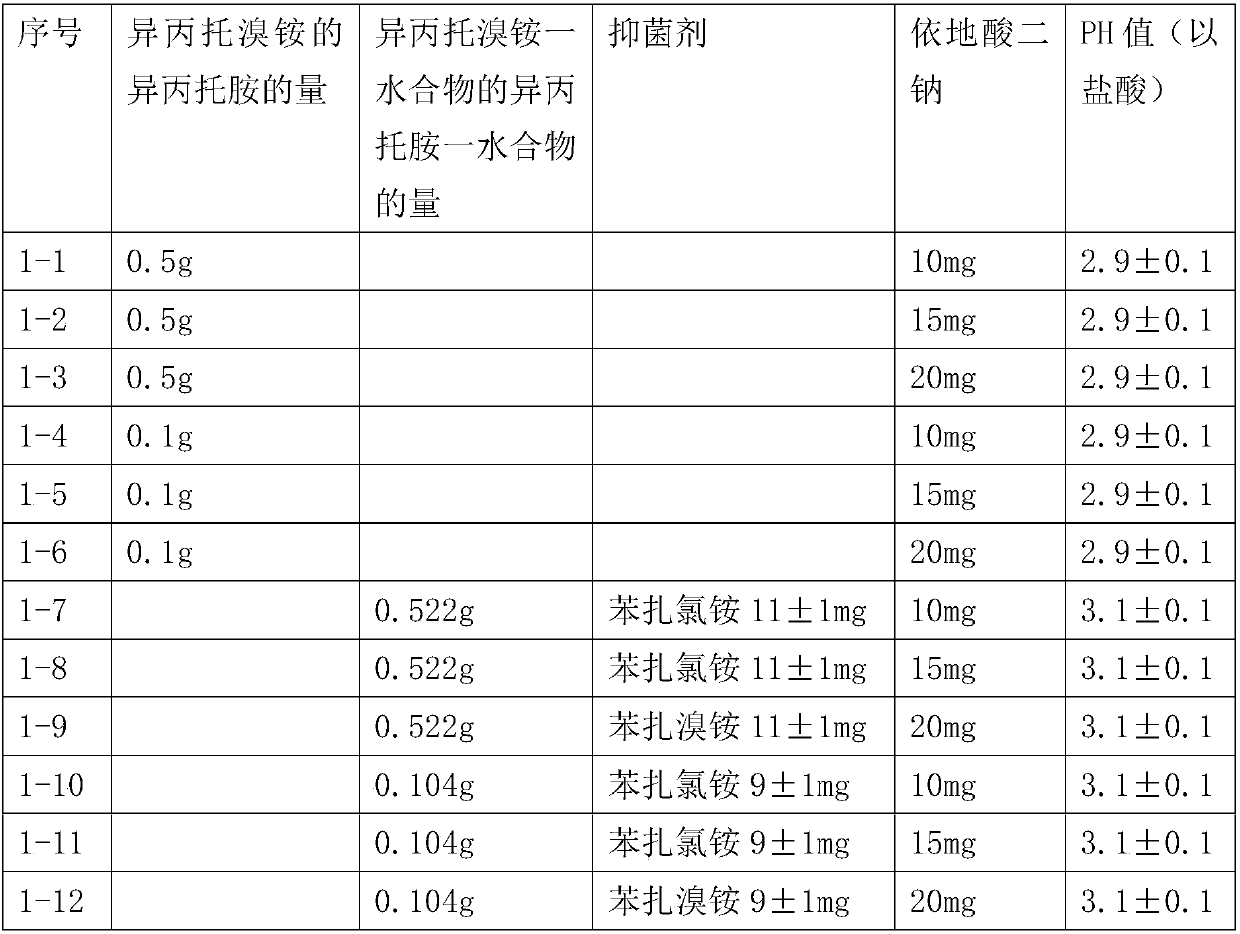
[0029] Prescription of pharmaceutical preparation of 100ml:
[0030]
[0031] Preparation method: Mix ipratropium bromide monohydrate or ipratropium bromide, bacteriostatic agent, edetate disodium and water to dissolve completely, and adjust the pH value with hydrochloric acid.
Embodiment 2
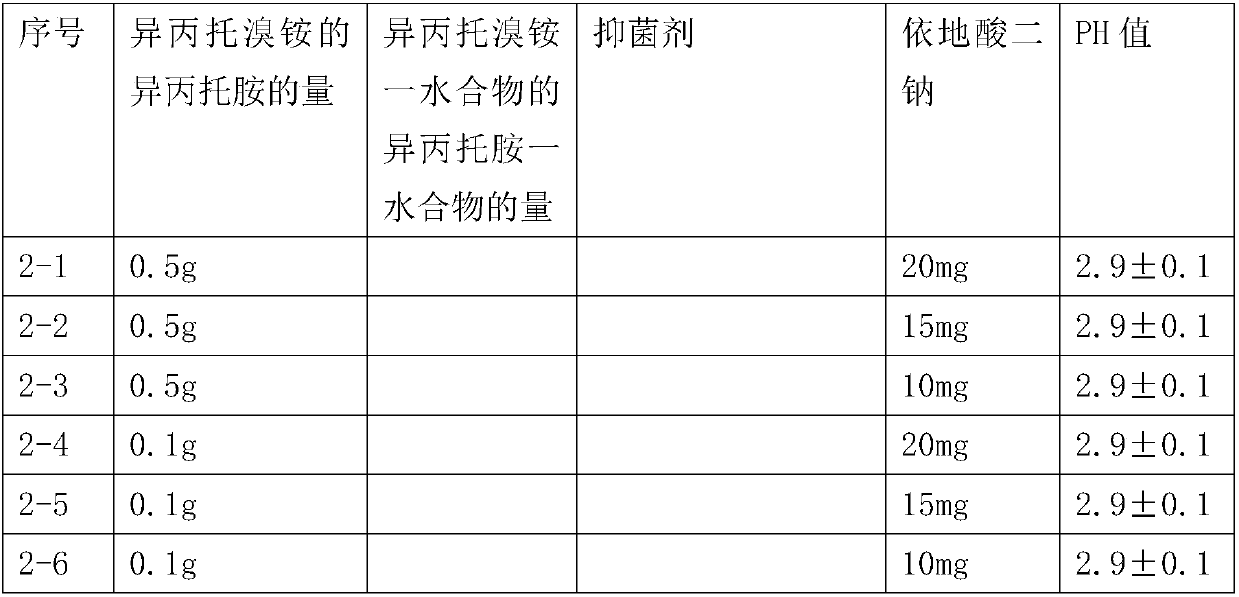
[0033] Prescription of pharmaceutical preparation of 100ml:
[0034]
[0035]
Embodiment 1
[0038] Prescription of pharmaceutical preparation of 100ml:
[0039]
[0040] The preparation method is the same as in Example 1.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to a drug ipratropium bromide for treating respiratory diseases, especially asthma, chronic obstructive pneumonia, bronchitis and the like, the drug is the inhalable aerosol without a propellant, and the invention also relates to a preparation method of the drug.
Description
Technical field: [0001] The present invention relates to the drug ipratropium bromide for treating respiratory diseases, especially asthma, chronic obstructive pneumonia, bronchitis, etc. The drug is an inhalable aerosol without propellant, and the present invention also relates to a preparation method of the drug . Background technique: [0002] Asthma is a chronic airway inflammation characterized by reversible airway obstruction and increased airway responsiveness. Airway obstruction is caused by two factors: increased secretions caused by bronchial mucosal inflammation, mucosal edema, and smooth muscle spasm stimulated by inflammation; while asthma Increased airway reactivity is also a result of damage to the bronchial epithelium due to airway inflammation. It is recognized that only by controlling the inflammation of the airway mucosa can the ultimate goal of reducing airway hyperresponsiveness and relieving asthma symptoms be achieved. At present, the drugs for the t...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): A61K9/72A61K31/46A61K47/18A61P11/00A61P11/06
CPCA61K9/0078A61K9/12A61K31/46A61K47/183A61P11/00A61P11/06
Inventor 张杰孙亮
Owner TIANJIN JINYAO GRP