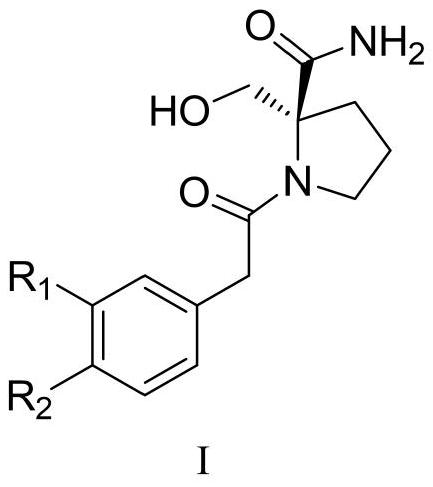
Preparation method of N-phenylacetyl-2-hydroxymethylpyrrolidine-2-formamide and medicinal application of N-phenylacetyl-2-hydroxymethylpyrrolidine-2-formamide
A technology of hydroxymethylpyrrolidine and benzyloxymethylpyrrolidine, which is applied in the direction of medical preparations containing active ingredients, antipyretics, drug combinations, etc., and can solve problems such as brain tissue damage
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
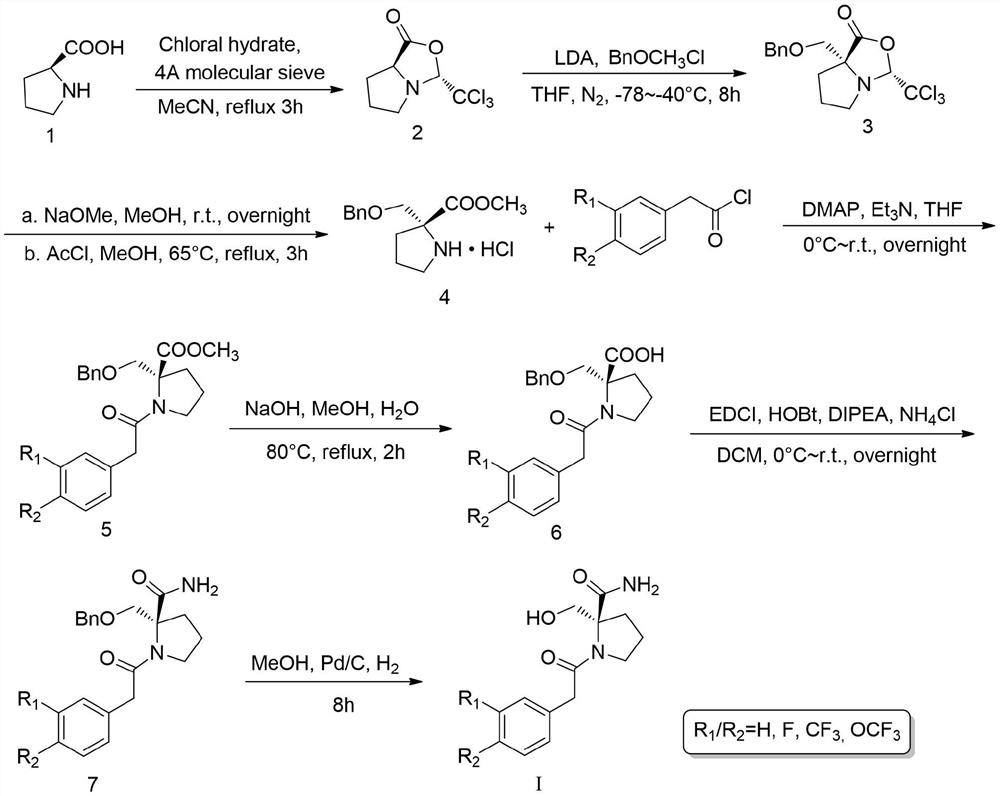
Examples
Embodiment 1
[0032] Embodiment 1 prepares oxazolidinone
[0033] Add 5.0g L-proline and 7.9g chloral hydrate (1.1eq) in 250mL single-necked flask, 15.6g 4A molecular sieve (2eq, water absorption is by 20% by weight) is dissolved in 80ml MeCN, is added dropwise to In the reaction system, reflux at 85°C for 3 hours after the dropwise addition, and TLC detected that the reaction was complete, filtered and evaporated to dryness, and used a mixed eluent of petroleum ether: ethyl acetate = 3:1 (containing 1.5% triethylamine) Separation and purification by column chromatography gave 7.3 g of white solid with a yield of 69%.
Embodiment 2
[0034] Embodiment 2 prepares α-benzyloxymethyl oxazolidinone
[0035] Add 5.0ml of diisopropylamine (1.2eq) and 50ml of anhydrous THF to a 250ml single-necked flask, put the flask into a low-temperature reactor and cool it to -78°C, and add 22.4ml of 1.6M n-butyllithium dropwise under nitrogen protection Hexane solution (1.2eq), reacted for 0.5 hours to prepare LDA.
[0036] Dissolve 7.3g of oxazolidinone (2) in 30ml of anhydrous THF, and add it dropwise into the above-mentioned LDA reaction flask, stir for 0.5 hours, add 6.2ml of chloromethyl benzyl ether (1.5eq) dropwise, and heat up to -40°C , and continued to stir for 8 hours, and nitrogen protection was required throughout the reaction. After the completion of the TLC detection reaction, add water to quench the reaction, extract with DCM and evaporate to dryness, use sherwood oil: ethyl acetate=30:1 eluent (containing 1.0% triethylamine) column chromatography to obtain 4.3g yellow liquid , yield 39.3%.
Embodiment 3
[0037] Embodiment 3 prepares α-benzyloxymethyl proline methyl ester hydrochloride
[0038] Add 850 mg of α-benzyloxymethyl oxazolidinone (3) and 30 ml of anhydrous methanol into a 100 ml one-necked flask, stir for a while, then add 50 mg of sodium metal (1 eq), and stir overnight at room temperature. 3.3ml of acetyl chloride (20eq) was added dropwise under ice-cooling, refluxed at 65°C for 3 hours, filtered and evaporated to dryness to obtain a yellow solid hydrochloride with a yield of 95.3%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More