Nucleoside compound and application thereof in treatment of feline infectious peritonitis
A compound and drug technology, applied in the field of nucleoside compounds, can solve the problems of large side effects and inability to reduce the high fatality rate of the disease
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0054] Embodiment 1: the preparation of compound 32:
[0055]
[0056] Step 1: Add 1.32 g of tert-butoxyacetic acid and 0.84 g of sodium bicarbonate to 30 mL of DMSO, and stir at room temperature for 20 min until dissolved. Add 2.0 g of raw material A1 at room temperature, raise the temperature to 65°C and react overnight, the raw material disappears and the product is formed. The reaction was stopped, extracted with EA / water until the aqueous layer was almost free of products, the organic phase was washed with saturated brine, dried, concentrated and put on the column, eluted with DCM / MeOH=30 / 1 to obtain 1.1 g of white solid B1.
[0057] 1 H NMR (400MHz, DMSO-d 6 )δ8.05–7.86(m,3H),6.92(d,J=4.4Hz,1H),6.82(d,J=4.4Hz,1H),6.36(d,J=6.0Hz,1H),5.42( d,J=5.8Hz,1H),4.68(t,J=5.4Hz,1H),4.38(d,J=9.8Hz,1H),4.28–4.14(m,2H),4.00(s,2H), 3.96–3.89(m,1H),1.12(s,9H).
[0058]Step 2: Dissolve 500 mg of raw compound B1 in 10 mL of DCM, add 3 mL of TFA dropwise under ice-cooling, and react...
Embodiment 2
[0069] Embodiment 2: solubility test
[0070] Experimental method: Determination with reference to high performance liquid chromatography (Chinese Pharmacopoeia 2020 Edition Sibu General Rules 0512).
[0071] a. Take about 1-2mg of the compound, put it in a 10ml measuring bottle, add 1ml of water, shake vigorously for 30 seconds every 5 minutes, and observe for 30 minutes. The test solution.
[0072] b. Take another appropriate amount of the reference substance, dissolve it in a solvent (DMSO: methanol = 10:90) and quantitatively dilute it to make a solution containing 0.1 mg per 1 ml, as the reference substance solution.
[0073] c. Precisely measure 20 μl each of the reference substance and the test solution, inject them into the liquid chromatograph respectively, and calculate the peak area according to the external standard method. The obtained data are shown in Table 2. Calculation formula: In the formula: A 供 : the main peak area of the test solution; A 对 : main ...
Embodiment 3
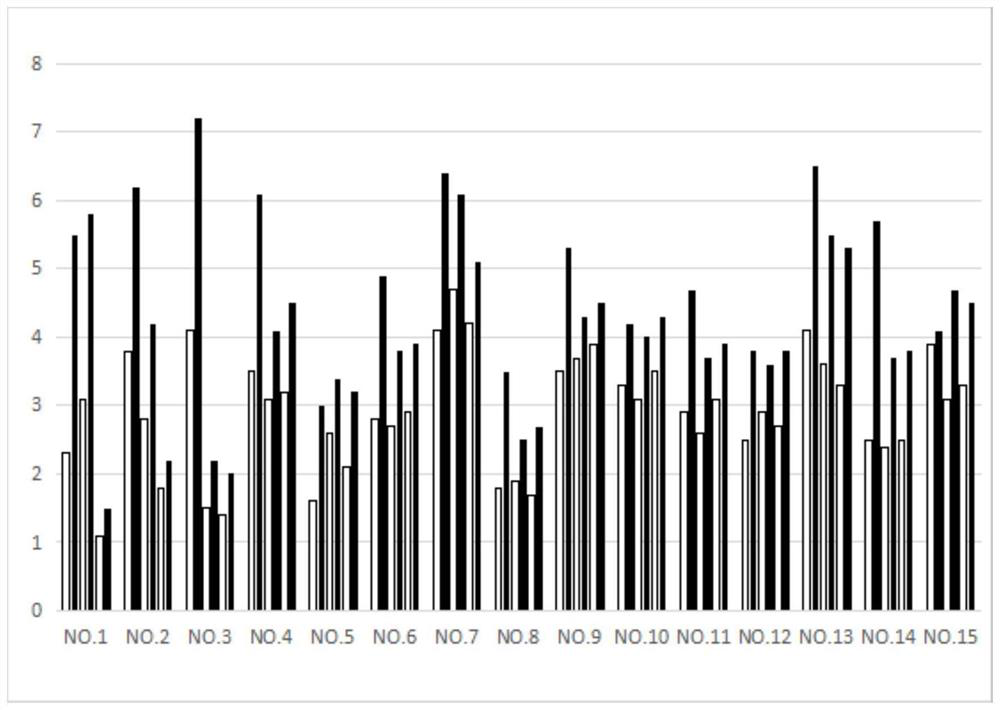
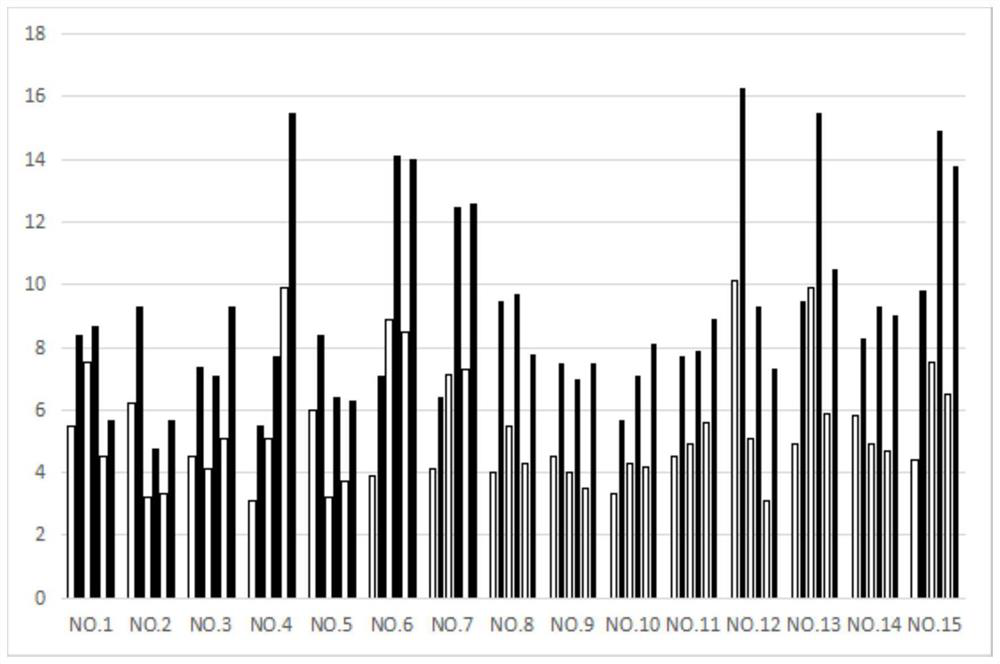
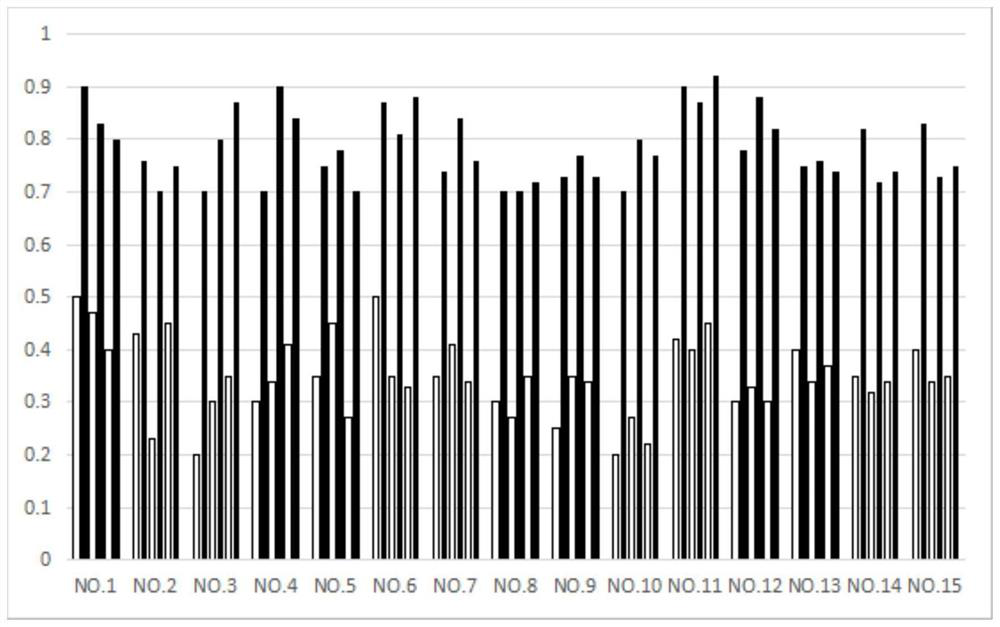
[0080] Embodiment 3: the drug effect experiment of the feline infectious peritonitis of compound 1,32,85
[0081] experimental method:
[0082] 15 mg of Compounds 1, 32, and 85 were respectively dissolved in 5 mL of solution (water:propylene glycol:PEG=2:1:1) to prepare injection solutions for injection administration. The injection can be stored at room temperature, use a 2mL disposable veterinary syringe to draw the corresponding volume of the injection solution, and remove the air upward. When injecting, choose a site with soft skin, loose subcutaneous tissue and fewer blood vessels, such as the neck or the medial thigh for subcutaneous injection. The dosage is 2 mg / Kg body weight once a day, blood is collected every three days and the blood routine and corresponding biochemical indicators are measured. The biochemical indicators of the cat return to normal as the end of the experiment. The index data measured in the experiment are body weight data and white blood cell dat...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More