Mesoderm and definitive endoderm cell populations
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Materials and Methods
[0077] ES cell growth and differentiation. ES cells were maintained on irradiated embryonic feeder cells in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 15% fetal calf serum (FCS), penicillin, streptomycin, LIF (1% conditioned medium) and 1.5×10−4 M monothioglycerol (MTG; Sigma). Two days prior to the onset of differentiation, cells were transferred on gelatinized plates in the same media. For the generation of EBs, ES cells were trypsinized and plated at various densities in differentiation cultures. Differentiation of EBs was carried out in 60 mm petri grade dishes in IMDM supplemented with 15% FCS, 2mM L-glutamine (Gibco / BRL), transferrin (200 ug / ml), 0.5mM ascorbic acid (Sigma), and 4.5×10−4 M MTG. Cultures were maintained in a humidified chamber in a 5% CO2 / air mixture at 37° C.
[0078] Serum Free Medium. Two different serum-free media were used in different aspects of the following examples: IIMD supplemented with Knockout SR (Gibco BRL) and S...
example 2
Generation of Targeted ES Cells
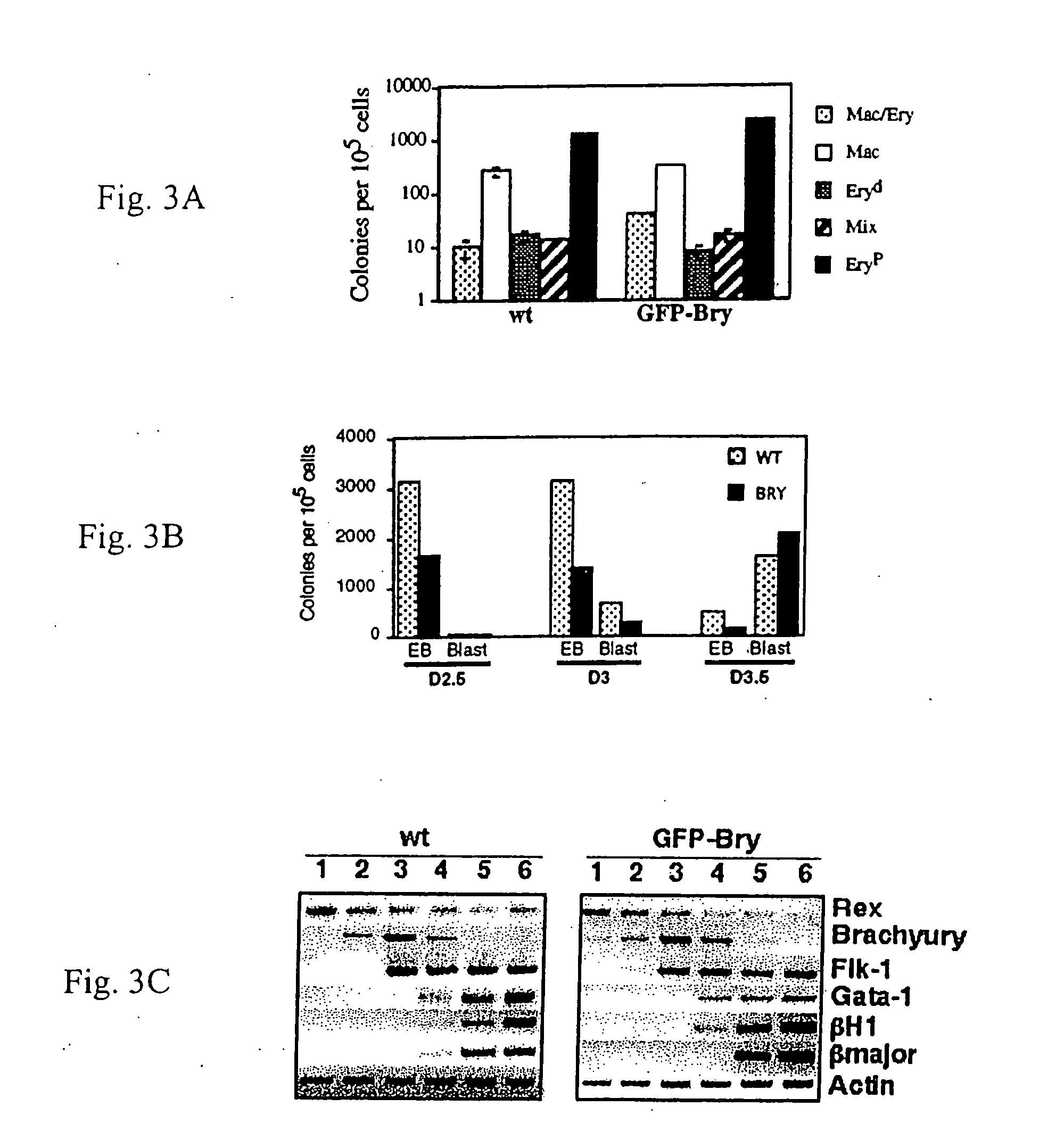
[0084] Under appropriate conditions in culture, embryonic stem (ES) cells will differentiate and form three dimensional colonies known as embryoid bodies (EBs) that contain developing cell populations from a broad spectrum of lineages. Smith (2001) Annu. Rev. Cell Dev. Biol. 17:435-62. Among these EB-derived populations, one can detect mesodermal derivatives including those of the hematopoietic, endothelial, cardiac muscle and skeletal muscle lineages.
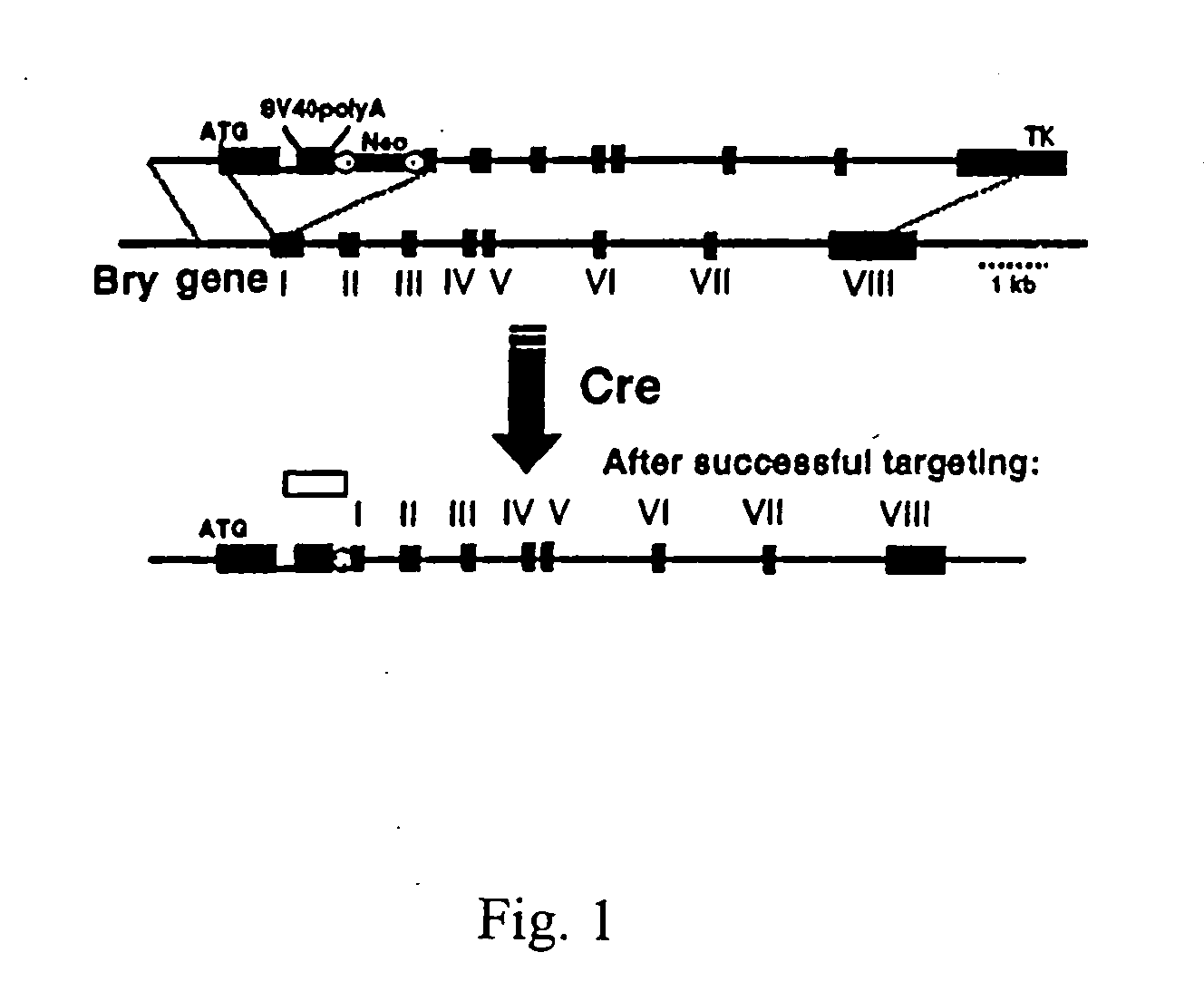
[0085] In order to track the onset of mesoderm in EBs and to isolate cells representing this population, the green fluorescence protein (GFP) was targeted to the brachyury locus. The targeting construct contained the GFP cDNA, and artificial intron, SV40 poly(A) sequences and a loxP flanked neo cassette in the first exon and is depicted in FIG. 1. The thymidine kinase (TK) gene was included at the 3′ end of the targeting construct to select against random integration. The targeting vector was constructe...
example 3
Isolation of Brachyury+ Cells
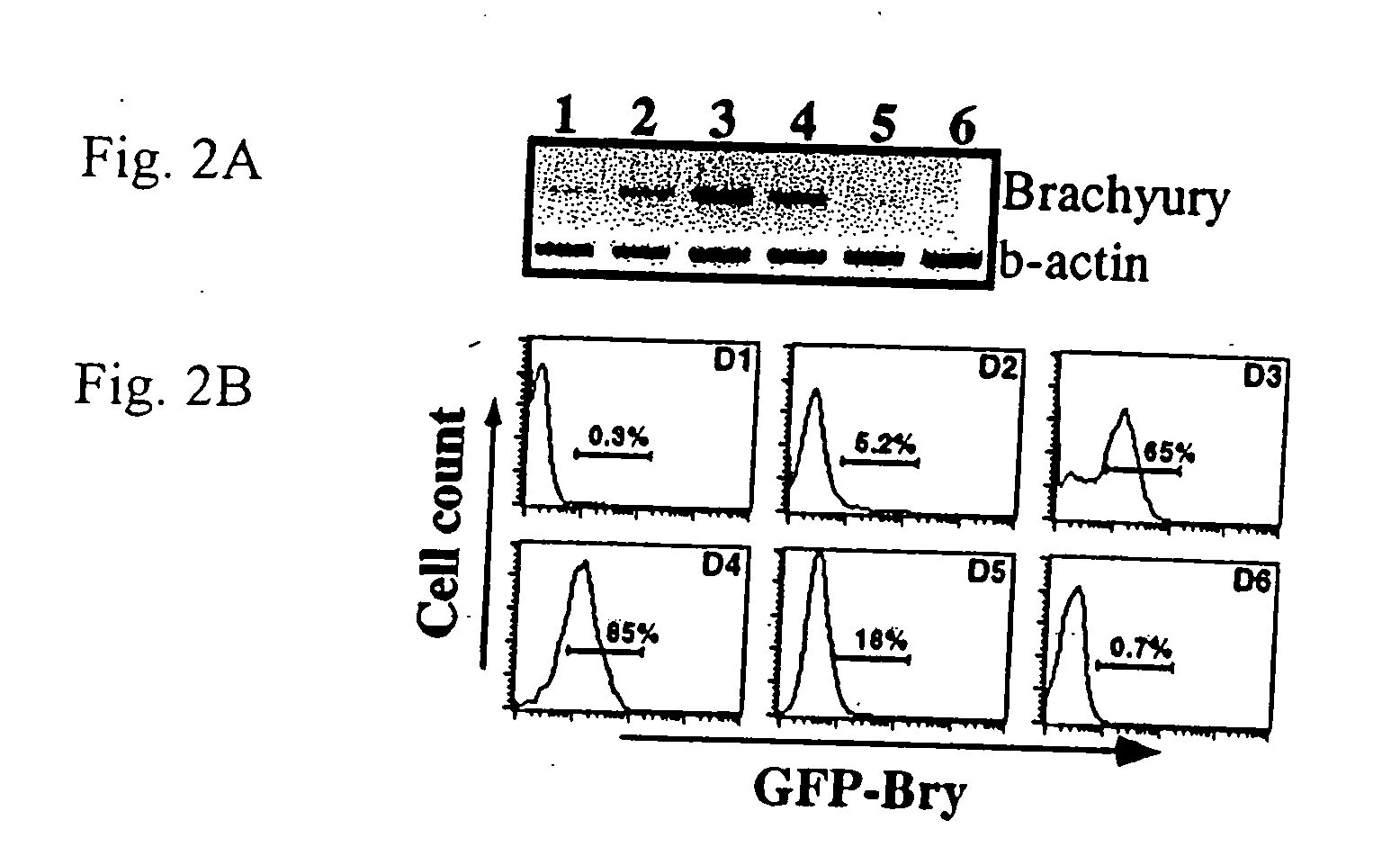
[0093] To determine if brachyury+ cells could be isolated based on GFP expression, the GFP+ population from day 3.5 EBs was sorted and analyzed for expression of appropriate genes. FIG. 4A shows the gates used for the isolation of the GFP positive (2) and negative(1) populations. RT-PCR analysis revealed brachyury expression was restricted to the GFP+ fraction indicating that cell sorting based on GFP expression can be used to isolate brachyury+ cells. Flk-1, one of the earliest makers of hematopoietic and endothelial development, was present at higher levels in GFP+ that in the GFP− fraction indicating that it was expressed in at least a subpopulation of brachyury+ cells. In contrast to brachyury and Flk-1, Pax-6, a gene involved in early neuronal development [79,80], was expressed at higher levels in the GFP− fraction consistent with precursors of this lineage being brachyury negative. These cell-sorting studies indicate that expression of GFP under t...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More